Translate this page into:
Correlation of patch test with repeated open application test in patients with suspected hair-dye dermatitis
Correspondence Address:
Sushruta Kathuria
Department of Dermatology and Venereology, Safdarjung Hospital and Vardhaman Mahavir Medical College, New Delhi
India
| How to cite this article: Khambra A, Ramesh V, Kathuria S. Correlation of patch test with repeated open application test in patients with suspected hair-dye dermatitis. Indian J Dermatol Venereol Leprol 2020;86:733-736 |
Sir,
Repeated open application test (ROAT) is an alternative method to patch test for detection of allergic contact dermatitis. It simulates the exposure pattern to allergen as in daily life, is cheaper, easier to perform and products can be tested as a whole. Hence, we proposed to compare ROAT with patch test as a tool to detect allergic contact dermatitis to hair dye in the Department of Dermatology and STD, Safdarjung Hospital and Vardhaman Mahavir Medical College, New Delhi. The study was approved by the ethical committee of the hospital. After informed consent, 50 patients (>12 years) having dermatitis over head and neck, trunk or upper limbs and temporal correlation with hair dye use were included in the study. Patients on oral corticosteroids or immunosuppressants and pregnant/lactating women were excluded. All patients were subjected to patch test and ROAT.
Patch test was done following standard guidelines as per International Contact Dermatitis Research Group (ICDRG) using Indian standard series (Systopic Laboratories, New Delhi). Reading taken on day 4 was considered as final.[1]
To perform ROAT, patients were asked to apply a one-fourth fingertip unit of all brands of hair dye used by them and vaseline (negative control) on a marked area of 3 × 3 cm on the volar aspect of the forearm for one week, as standardized by Hannuksela and Salo.[2] Readings were recorded as per Johansen's modified scale.[1] Ten healthy controls with no history of contact dermatitis or atopy were also asked to perform ROAT with two different commonly used brands of hair dye (Garnier and Godrej) and vaseline as control. The data was analyzed with Statistical Package for Social Sciences (SPSS) version 21.0.
Sixty-two patients presented with suspected hair-dye dermatitis, of which 50 completed the study. The clinical characteristics and hair dye used by the patients are given in [Table - 1]. On patch test, 36/50 (72%) showed positive reaction to paraphenylenediamine (PPD) [Figure - 1] along with other allergens [Table - 2]. In those who had positive reaction to PPD and other allergens such as parthenium, nickel sulphate, nitrofurazone, thiuram mix, fragrance mix, colophony and mercaptobenzothiazole, only PPD was found clinically relevant [Table - 2]. The diagnosis in 14 patients who tested negative on patch test and ROAT as well was revised based on repeat history and patch test results [Table - 2] to parthenium in 5, nickel dermatitis in 3, nitrofurazone in 3, colophony in 1 and fragrance mix in 1 and endogenous dermatitis in 1.

 |
| Figure 1: Patch test showing bullous reaction to paraphenylenediamine on day 4 |
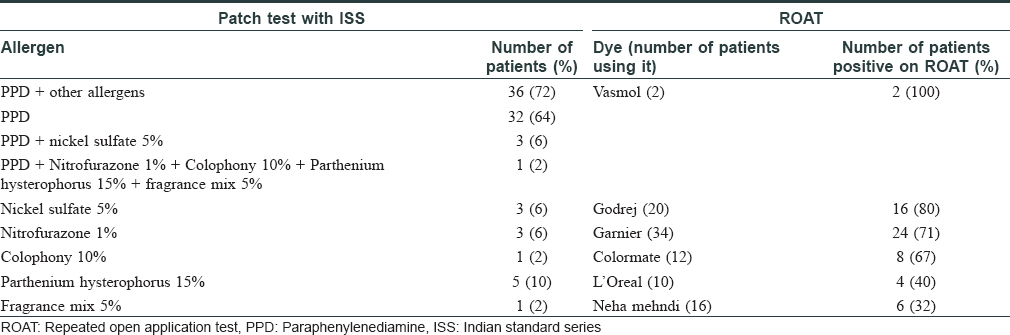
Thirty (60%) patients showed positive reaction on ROAT [Table - 2] and [Figure - 2] and all were patch test positive too. In the 30 ROAT-positive patients, there were 60 positive reactions to different dyes of which 49 occurred on day 2, and the remaining 11 occurred on day 4. No new positive reaction appeared on day 7. On comparing ROAT with patch test, correlation coefficient was 0.846 and P value was < 0.0001. Taking patch test as the standard, sensitivity of ROAT was 83.33% (95% CI: 67.19- 93.63%), specificity was 100% (95% CI: 76.84-100.00%), positive predictive value was 100% and negative predictive value was 70%. ROAT was negative in all controls. On comparison with 50 cases, P value was highly significant (<0.001). No adverse effects to patch test or ROAT were noted.
 |
| Figure 2: Repeated open application test showing positive reaction and kissing lesions on day 2 |
PPD is the most common contact sensitizer in hair-dye dermatitis, others being resorcinol, henna, lead acetate, m-aminophenol, o-aminophenol, p-aminophenol and toluene-2,5-diamine sulfate. The permissible limit of PPD in hair dyes is 6% and estimated PPD sensitivity in general population is around 1%.[3] Even in low concentration, PPD is a potent contact sensitizer as seen on EC3 value (effective concentration of the test substance required to produce a three-fold increase in the stimulation index) for PPD on local lymph node assay.[4] The reported prevalence of positive patch test reactions to PPD among dermatitis patients is 4.4% in Asia, 4.1% in Europe, 6.0% in North America, and 11.5% in India.[5],[6]
In India, patch test positivity in hair-dye dermatitis is 67.5% similar to our study (72%)[7] while in Korea and Japan, it is lower (25% and 35.1%, respectively), which could be because of other allergens such as toluene-2,5-diamine, p-aminophenol, m-aminophenol, cysteamine HCl and o-aminophenol, causing hair dye dermatitis in their population.[8],[9]
ROAT is used to determine the relevance of doubtful positive patch test reactions to preparations in which the suspected allergen is present in a low concentration. ROAT is as good as patch test in determining allergy to oxidized limonene[10] in high concentration, methyldibromoglutaronitrile[11] and nickel,[3] but is inferior to patch test for hydroxycitronellal, formaldehyde and chromium.[12]
The correlation coefficient between patch test and ROAT in our study was 0.84, suggesting significant correlation. Even though ROAT is inferior to patch testing in confirming contact dermatitis to hair dye, it can be considered as an alternative tool in situ ations where patch test cannot be conducted due to reasons such as remote area, non-availability of patch test kit, expertise to read the test, unavailability of PPD antigen or non-compliance by patient.
ROAT has a theoretical risk of sensitization to PPD in patients who are not allergic to it as the concentration of PPD in dyes is high but there are no guidelines for not performing or discouraging ROAT in already sensitized patients. Since ROAT is not performed under occlusion, the risk of sensitization maybe not as high as suspected. The disadvantage of using hair dye 'as is' for ROAT is that it does not give information on which antigen/constituent caused the allergy. Unavailability of hair dye allergens other than PPD and the small number of patients were the limitations of this study.
ROAT is less sensitive than patch test, but highly specific with a positive predictive value of 100% and negative predictive value of 70%. ROAT correlates well with patch test and can be used as an alternative tool for diagnosis only in situ ations where patch test cannot be performed.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patients have given their consent for their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Johansen JD, Aalto-Korte K, Agner T, Andersen KE, Bircher A, Bruze M, et al. European Society of Contact Dermatitis guideline for diagnostic patch testing – Recommendations on best practice. Contact Dermatitis 2015;73:195-221.
[Google Scholar]
|
| 2. |
Hannuksela M, Salo H. The repeated open application test (ROAT). Contact Dermatitis 1986;14:221-7.
[Google Scholar]
|
| 3. |
de Groot AC. Contact allergy to cosmetics: Causative ingredients. Contact Dermatitis 1987;17:26-34.
[Google Scholar]
|
| 4. |
Thyssen JP, Søsted H, Uter W, Schnuch A, Giménez-Arnau AM, Vigan M, et al. Self-testing for contact sensitization to hair dyes – Scientific considerations and clinical concerns of an industry-led screening programme. Contact Dermatitis 2012;66:300-11.
[Google Scholar]
|
| 5. |
Thyssen JP, White JM; European Society of Contact Dermatitis. Epidemiological data on consumer allergy to p-phenylenediamine. Contact Dermatitis 2008;59:327-43.
[Google Scholar]
|
| 6. |
Sharma VK, Chakrabarti A. Common contact sensitizers in Chandigarh, India. A study of 200 patients with the European standard series. Contact Dermatitis 1998;38:127-31.
[Google Scholar]
|
| 7. |
Gupta M, Mahajan VK, Mehta KS, Chauhan PS. Hair dye dermatitis and p-phenylenediamine contact sensitivity: A preliminary report. Indian Dermatol Online J 2015;6:241-6.
[Google Scholar]
|
| 8. |
Lee JY, Kim CW, Kim SS. Analysis of the results from the patch test to para-phenylenediamine in the TRUE test in patients with a hair dye contact allergy. Ann Dermatol 2015;27:171-7.
[Google Scholar]
|
| 9. |
Ito A, Nishioka K, Kanto H, Yagami A, Yamada S, Sugiura M, et al. A multi-institutional joint study of contact dermatitis related to hair colouring and perming agents in Japan. Contact Dermatitis 2017;77:42-8.
[Google Scholar]
|
| 10. |
Bennike NH, Palangi L, Christensson JB, Nilsson U, Zachariae C, Johansen JD, et al. Allergic contact dermatitis caused by hydroperoxides of limonene and dose-response relationship-A repeated open application test (ROAT) study. Contact Dermatitis 2019;80:208-16.
[Google Scholar]
|
| 11. |
Fischer LA, Johansen JD, Menné T. Methyldibromoglutaronitrile allergy: Relationship between patch test and repeated open application test thresholds. Br J Dermatol 2008;159:1138-43.
[Google Scholar]
|
| 12. |
Villarama CD, Maibach HI. Correlations of patch test reactivity and the repeated open application test (ROAT)/provocative use test (PUT). Food Chem Toxicol 2004;42:1719-25.
[Google Scholar]
|
Fulltext Views
5,372
PDF downloads
1,179





