Translate this page into:
A randomized, open-label, comparative study of oral tranexamic acid and tranexamic acid microinjections in patients with melasma
2 Department of Preventive and Social Medicine, Hindu Rao Hospital, New Delhi, India
Correspondence Address:
Rachita R Misri
2nd Floor OPD Building, Department of Dermatology, Venereology and Leprosy, Hindu Rao Hospital, New Delhi - 110 007
India
| How to cite this article: Khurana VK, Misri RR, Agarwal S, Thole AV, Kumar S, Anand T. A randomized, open-label, comparative study of oral tranexamic acid and tranexamic acid microinjections in patients with melasma. Indian J Dermatol Venereol Leprol 2019;85:39-43 |
Abstract
Background: Melasma poses a great challenge as its treatment modalities are unsatisfactory. Treatment using tranexamic acid is a novel concept.
Aim: This study aimed to compare the therapeutic efficacy and safety of oral tranexamic acid and tranexamic acid microinjections in patients with melasma.
Methods: This is a prospective, randomized, open-label study with a sample size of 64, 32 in each treatment arm. Thirty-two patients were administered localized microinjections (4 mg/ml) of tranexamic acid monthly in 1 arm, while in the other arm, 32 were given oral tranexamic acid 250 mg twice a day. Patients were followed up for 3 consecutive months. Clinical photographs were taken at each visit, and a modified melasma area and severity index scoring was performed at the beginning and end of treatment.
Results: Improvement in melasma area and severity index score in the oral group was 57.5% as compared to 43.5% in the intralesional group. All 32 patients in the oral group (100%) showed >50% improvement, out of which 8 showed >75% improvement. In the intralesional group, 17 (53%) patients had >50% improvement, of which 3 had >75% improvement. The remaining 15 patients in this group had <50% improvement. Thus, the oral group showed a more significant response as compared to the intralesional group. No major adverse effects were observed in both the groups. At 6-month follow-up, two patients (6.2%) in the oral group had recurrence as compared to three patients (9.4%) in the intralesional group.
Limitations: A small sample size was one of the limitations in this study. The dose of tranexamic acid in microinjections and the frequency of injections could have been increased.
Conclusion: Tranexamic acid provides rapid and sustained improvement in the treatment of melasma. It is easily available and affordable. Oral route is undoubtedly efficacious, but the results of microinjections, while encouraging, can probably be enhanced by either increasing the frequency of injections or increasing the concentration of the preparation.
Introduction
Melasma is a common acquired dermatosis characterized by the presence of light-to-dark brown macules and patches involving the sun-exposed areas of the face and neck. This disease is commonly observed in women.[1] Although the precise cause of melasma is still unknown, some commonly identified contributing factors are genetic susceptibility, ultraviolet light exposure, pregnancy, sex hormones, contraceptive pills, thyroid disease, cosmetics and phototoxic drugs.[2]
Various treatment modalities are used for melasma. These include the use of sunscreens, hypopigmenting agents, superficial peeling agents such as glycolic and lactic acid and laser therapy.[1],[3],[4],[5] Newer studies have found that tranexamic acid, a hemostatic agent, has hypopigmentary effect on melasma lesions and also prevents ultraviolet-induced pigmentation.[2],[6],[7] In addition to its hemostatic effects, tranexamic acid exhibits anti-allergic and anti-inflammatory effects on various skin diseases such as angioedema.[8] Its anti-inflammatory property appears to be related to its inhibitory effect on melanogenesis.[6],[8] Tranexamic acid is reported to improve melasma when injected locally or used in its oral and topical forms.
Although several treatment modalities are available, management of this condition is a challenging task for doctors and the results are frequently unsatisfactory, with a high recurrence. However, there are many ongoing studies aiming to find more effective and safe treatments.
Methods
A prospective randomized open-label study was conducted on 64 clinically diagnosed melasma patients. Sample size was calculated to be a minimum of 14 in each group at alpha error = 5% and power 80% at 95% confidence interval. Sample size was calculated using the study as shown in [Table - 1].

Patients were selected from among those coming for treatment of various dermatoses in our outpatient department during 1-year period of May 2014–May 2015, after obtaining approval from the Institutional Ethical Committee.
This study included adult men and women with melasma between the age group of 20 and 50 years. Patients with a history of bleeding disorders, use of oral anticoagulant drugs or any other photosensitizing drugs such as nonsteroidal anti-inflammatory drugs, tetracycline, spironolactone, phenytoin, carbamazepine, known allergy to tranexamic acid, other concomitant medical history and the use of other depigmenting oral or topical agents in the past 1 month were exclusion criteria. Additionally, females with a history of pregnancy/lactation or use of oral contraceptive pills/hormonal replacement therapy at the time of or during the past 12 months were excluded from the study. Patients with a history of any treatment for melasma in the past 6 weeks in any form were also excluded from the study.
Patients were randomly selected for oral and topical therapy for melasma. Randomization was done by random number allocation. After obtaining written informed consent, history was taken and clinical examination done. The lesion was also examined under a Woods lamp. Baseline investigations of liver and renal functions and a coagulation profile were done, and repeated after 3 months. Modified melasma area and severity index scoring system [Figure - 1] was used to assess the severity of melasma and to compare the response in each patient with the initial score to find the percentage improvement. Patients in the oral therapy group were given tablet tranexamic acid (250 mg) twice daily for 3 months. In the intralesional group, patients were administered intradermal injections of tranexamic acid. About 2 U of tranexamic acid was drawn in a 40 U/ml 30-gauge insulin syringe and diluted with normal saline up to 1 ml (remaining 38 U out of total 40 U) to get a concentration of approximately 4 mg/ml of tranexamic acid.[9] Intradermal injections were given at the site of melasma, after application of topical anesthetic, keeping a distance of around 1 cm from each injection, with a maximum of 8 mg in a single sitting. Three such sessions at intervals of a month were carried out. Various measures for strict photoprotection were explained to each patient, for better and sustained improvement. All patients were given the same sunscreen (of sun protection factor 50) for the entire 6 months.
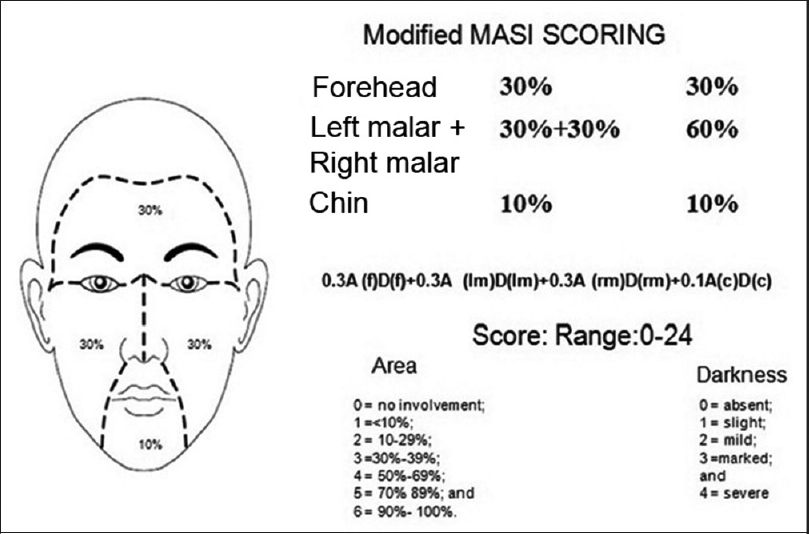 |
| Figure 1: Modified melasma area and severity index scoring |
To assess response, clinical photographs were taken at each visit; modified melasma area and severity index scores were calculated at the beginning and end of the therapy. Subjective response to treatment, according to patient, was graded at the end of the study as follows: no response, no improvement; mild response, <25% improvement; moderate response, 25%–< 50% improvement; good response, 50%–< 75% improvement and excellent response, >75% improvement. Final response was evaluated according to the melasma area and severity index scores and subjective patient response. Any complications and side effects were also noted during these follow-ups. For another 3 months after treatment, the patients were examined at monthly intervals to look for any relapse or complication.
Histopathology could not be done due to apprehension of patients and as it involved the face. Dermoscopy was not available and no previous studies have used it as a tool to assess response to tranexamic acid.
Statistical analysis was done using SPSS software version 20 (IBM Statistical package for the Social Sciences. IBM SPSS Statistics for Windows, Version 20.0. Armonk, NY: IBM Corp.). The difference in mean melasma area and severity index scores in each group was calculated using paired t-test. The difference in mean melasma area and severity index scores between the two groups was calculated using Chi-square test using 2 × 2 table.
Results
Out of 72 patients enrolled in the study, 64 (88.8%) successfully completed the trial. Forty-seven (65.27%) patients belonged to the age group of 30–50 years. The number of women was more (54) compared to men (10) in both the study groups [Table - 2]. All the patients had skin type 4 or 5 [Table - 3]. Forty-four (68.8%) patients had mixed pattern of melasma, 17 (26.6%) had epidermal and 3 (4.7%) had dermal [Table - 3]. The distribution of melasma was either centrofacial or malar [Table - 3]. None of the patients were on any drugs.


Mean melasma area and severity index score of the patients at the beginning and end of the trial with percentage improvement is shown in [Table - 4] and graphically in [Figure - 2]. Mean decrease in melasma area and severity index score in oral group was significantly higher than in the microinjection group (Mann–Whitney U-test: P < 0.001, df = 62). Improvement in oral group was 57.5% with P < 0.01 (paired t-test: 12.111). In intralesional group, the improvement was 43.6% with P value of 0.047 (paired t-test: 2.068).

 |
| Figure 2: Graph showing comparison of decrease in melasma area and severity index score in oral and intralesional groups |
All the 32 patients in the oral group (100%) showed >50% improvement, of which 8 showed >75% improvement. In the intralesional group, 17 (53%) patients had >50% improvement, 3 of whom had >75% improvement. The remaining 15 patients in this group had <50% improvement [Table - 5]. Thus, the oral group showed significantly better response as compared to the intralesional group (2 × 2 table; χ2 = 19.592, df < 1. P <0.01).

[Figure - 3] and [Figure - 4] show examples of clinical improvement in some of the patients.
 |
| Figure 3: Patient treated with oral tranexamic acid – before and after treatment |
 |
| Figure 4: Patient treated with intralesional tranexamic acid – before and after treatment |
In the oral group, the difference in the mean fall in melasma area and severity index score in epidermal type was 2.92 (standard deviation ±0.92) and in mixed type was 4.55 (standard deviation ±2.06). Although the fall in mixed type was more, it did not differ significantly (P = 0.058 by Mann–Whitney U-test). In the intralesional group, the mean fall in melasma area and severity index in epidermal type was 3.12 (standard deviation ±4.99) and in mixed type was 1.01 (standard deviation ±4.81). Here too, the fall was not statistically significant (P = 0.31 by Mann–Whitney U-test).
No major side effects were observed except for mild pain and erythema in the intralesional group patients, which lasted for 2–3 days. In the oral group, two patients(6.25%) had gastritis and one patient (3.12%) had oligomenorrhea at the end of 3 months. There was no change in coagulation parameters at the end of 3 months.
At 6 months of follow-up, 6.25% in the oral group had recurrence with darkening of the lesions and in the intralesional group 9.37% had repigmentation of lesions at the end of 6 months.
Discussion
Tranexamic acid (trans-4-aminomethylcyclohexanecarboxylic acid) is a plasmin inhibitor used to prevent abnormal fibrinolysis to reduce blood loss.[10] Ultraviolet irradiation induces plasminogen activator synthesis and plasmin activity in cultured keratinocytes. Plasmin-activated precursors of secretory phospholipase A2 which participates in the production of arachidonic acid from membrane phospholipids is a precursor to prostaglandins E2 and leukotrienes which can lead to melanogenesis.[11] Plasmin also leads to release of basic fibroblast growth factor which is a potent melanocyte growth factor. Hence, tranexamic acid prevents binding of plasminogen to keratinocyte which results in less arachidonic acid and diminished ability to produce prostaglandins and subsequently reduces melanogenesis in melanocyte.[12] By injecting intradermally, it may be possible to treat the dermal and mixed types of melasma.
Limited studies are found in literature regarding the use of tranexamic acid in melasma. In 1979, Nijor was the first to study and report on the action of tranexamic acid in melasma.[13] In 1985, Hajime et al. showed that forty patients aged 20–60 years had their melasma reduced in severity with 1–1.5 g daily oral tranexamic acid in 10 weeks' time.[14] In 1988, 11 patients with melasma were treated with oral tranexamic acid 0.75–1.5 g/day.[15] All had decreased severity; however, pigmentation recurred after some months of cessation of therapy. Zhu and Yang[16] and Liu et al.[17] each gave 250 mg tranexamic acid 3 times a day along with Vitamin C and E for duration of 6–8 weeks. Both the groups showed significant reduction in pigmentation with tolerable gastrointestinal symptoms and no abnormality in coagulation parameters. They also found that increasing the treatment duration was more effective than increasing the dose of tranexamic acid. In 2008, Mafune et al. used 750 mg oral tranexamic acid two tablets 3 times daily.[18] In 2013 Cho et al. performed the first controlled trial by administering 500 mg/day as an adjuvant to patients treated with intense pulse light or neodymium-doped yttrium aluminum garnet laser, as compared to patients who received the same treatment without tranexamic acid.[19] Six-month treatment led to significantly higher improvement without any systemic side effects, in the tranexamic acid group.
The usual effective dose for melasma is 250–500 mg 2–3 times daily, which is much lower than the dose to reduce bleeding. Our study is in agreement with other many studies where successful lightening was observed following oral tranexamic acid administration, with some side effects of gastritis (6.25%) and oligomenorrhea (3.12%) which disappeared after cessation of therapy. Commonly reported side effects include nausea, diarrhea, orthostatic reactions, disturbances in color vision, occasionally anaphylactic shock and acute renal cortical necrosis. Thrombotic incidents have been reported at doses ranging from 1.5 to 6 g/day.[12] After 6-month follow-up period, recurrence was seen in two patients (6.25%) which could be effectively treated again using tranexamic acid therapy.
Localized microinjection, also known as mesotherapy, is a widely used technique in medicine. It aims at directly applying an adequate amount of medication at the given site thus avoiding oral medications. This allows lower dosage of drugs to be used. In our study, we used monthly intradermal injections of tranexamic acid on the affected area with significant decrease in melasma area and severity index scores with no significant side effects. In 2006, Lee et al. showed that 85 patients who completed weekly intradermal injections of tranexamic acid for 12 weeks had significant decrease in their melasma area and severity index scores.[2] Budamakuntla et al. compared weekly intradermal injections of tranexamic acid in one arm with microneedling with tranexamic acid in the second arm and found significant decrease in melasma area and severity index scores in both the groups.[9] There are no standard guidelines for giving intralesional injections of tranexamic acid for melasma. In our study, we gave monthly injections instead of weekly ones, as mentioned in the above two studies, to see if we get an appreciable response with this technique, since repeated injections are quite traumatic for the patient, due to multiple pricks and visits to the doctor. We got a good response of 43.6% which is statistically significant but this was less than the response achieved by the oral route.
We compared the safety and efficacy of treatment in both these methods of drug delivery. The melasma area and severity index scores showed a significant decrease in baseline to the end of 12th week in both the arms. The scores showed better improvement in patients on oral tranexamic acid than those receiving intralesional injections, and this was statistically significant. This can be attributed to lower dosages of the injected drug, as well as decreased frequency of injections. On comparing the differences between the fall in melasma area and severity index score between epidermal and mixed types of melasma, there was no difference, thus showing it to be equally effective in all types of melasma. All scores remained almost the same during the next 3 months of follow-up.
Based on our study, we suggest oral tranexamic acid 250 mg 2 times daily for at least 3 months. Prolonged prescription period should have a better effect rather than increasing the dose. Increasing the frequency of intralesional injections to once a week rather than a month would increase the efficacy. It would be also interesting to observe if increasing the concentration of intralesional tranexamic acid from 4 mg/ml would increase the efficacy of the results. Further studies with larger number of patients are required. On the basis of the above findings, tranexamic acid can be used as a safe effective therapeutic agent for the treatment of melasma. It is the only modality that can prevent the activation of melanocyte by sunlight, hormonal influence through the inhibition of plasminogen activator system.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patients have given their consent for their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Grimes PE. Melasma. Etiologic and therapeutic considerations. Arch Dermatol 1995;131:1453-7.
[Google Scholar]
|
| 2. |
Lee JH, Park JG, Lim SH, Kim JY, Ahn KY, Kim MY, et al. Localized intradermal microinjection of tranexamic acid for treatment of melasma in Asian patients: A preliminary clinical trial. Dermatol Surg 2006;32:626-31.
[Google Scholar]
|
| 3. |
Rendon M, Berneburg M, Arellano I, Picardo M. Treatment of melasma. J Am Acad Dermatol 2006;54 5 Suppl 2:S272-81.
[Google Scholar]
|
| 4. |
Prignano F, Ortonne JP, Buggiani G, Lotti T. Therapeutical approaches in melasma. Dermatol Clin 2007;25:337-42, viii.
[Google Scholar]
|
| 5. |
Gupta AK, Gover MD, Nouri K, Taylor S. The treatment of melasma: a review of clinical trials. J Am Acad Dermatol 2006;55:1048-65.
[Google Scholar]
|
| 6. |
Maeda K, Naganuma M. Topical trans-4-aminomethylcyclohexanec arboxylic acid prevents ultraviolet radiation-induced pigmentation. J Photochem Photobiol B 1998;47:136-41.
[Google Scholar]
|
| 7. |
Kondou S, Okada Y, Tomita Y. Clinical study of effect of tranexamic acid emulsion on melasma and freckles. Skin Res 2007;6:309-15.
[Google Scholar]
|
| 8. |
Maeda K, Tomita Y. Mechanism of the inhibitory effect of tranexamic acid on melanogenesis in cultured human melanocytes in the presence of keratinocyte-conditioned medium. J Health Sci 2007;53:389-96.
[Google Scholar]
|
| 9. |
Budamakuntla L, Loganathan E, Suresh DH, Shanmugam S, Suryanarayan S, Dongare A, et al. Arandomised, open-label, comparative study of tranexamic acid microinjections and tranexamic acid with microneedling in patients with melasma. J Cutan Aesthet Surg 2013;6:139-43.
[Google Scholar]
|
| 10. |
Shimizu M, Naito T, Okano A, Aoyagi T. Trans-4 aminomethylcyclohex anecarboxylic acid as a potent antiplasmin agent. Chem Pharm Bull (Tokyo) 1965;13:1012-4.
[Google Scholar]
|
| 11. |
Akashima A, Yasuda S, Mizuno N. Determination of the action spectrum for UV-induced plasminogen activator synthesis in mouse keratinocytes in vitro. J Dermatol Sci 1992;4:11-7.
[Google Scholar]
|
| 12. |
Tse TW, Hui E. Tranexamic acid: An important adjuvant in the treatment of melasma. J Cosmet Dermatol 2013;12:57-66.
[Google Scholar]
|
| 13. |
Nijor T. Treatment of melasma with tranexamic acid. Clin Res 1979;13:3129-31.
[Google Scholar]
|
| 14. |
Hajime M, Mineo T, Yoshio T. Oral administration therapy with tranexamic acid for melasma. Nishinihon J Dermatol 1985;47:1101-4.
[Google Scholar]
|
| 15. |
Higashi N. Treatment of melasma with oral tranexamic acid. Skin Res 1988;30:676-80.
[Google Scholar]
|
| 16. |
Zhu HJ, Yang XH. The clinical study of acidum tranex-amicum on melasma. Pharm Program 2001;3:178-81.
[Google Scholar]
|
| 17. |
Liu H, Kou CC, Yeung CW. Effectiveness of tranexamic acid in treating melasma and observation of its safety. Chin J Med Aesthet Cosmetol 2005;11:361-3.
[Google Scholar]
|
| 18. |
Mafune E, Morimoto Y, Iizuka Y. Tranexamic acid and melasma. Farmacia 2008;44:437-42.
[Google Scholar]
|
| 19. |
Cho HH, Choi M, Cho S, Lee JH. Role of oral tranexamic acid in melasma patients treated with IPL and low fluence QS Nd:YAG laser. J Dermatol Treat 2013;24:292-6.
[Google Scholar]
|
Fulltext Views
11,644
PDF downloads
3,792





