Translate this page into:
Strongyloides stercoralis hyperinfection: An often missed but potentially fatal cause of anemia and hypoalbuminemia in leprosy patients on long-term steroid therapy
2 Department of Pathology, All India Institute of Medical Sciences, New Delhi, India
Corresponding Author:
Neena Khanna
Department of Dermatology and Venereology, Teaching Block, All India Institute of Medical Sciences, Ansari Nagar, New Delhi - 110 029
India
neena_aiims@yahoo.co.in
| How to cite this article: Gupta V, Bhatia S, Mridha AR, Das P, Khanna N. Strongyloides stercoralis hyperinfection: An often missed but potentially fatal cause of anemia and hypoalbuminemia in leprosy patients on long-term steroid therapy. Indian J Dermatol Venereol Leprol 2017;83:381-383 |
Sir,
Strongyloides stercoralis can cause hyperinfection in immunosuppressed patients which can be fatal, if left untreated. Its nonspecific clinical features often lead to a missed or delayed diagnosis. We report a patient with lepromatous leprosy on long-term steroid therapy, who presented with anemia and generalized edema due to Strongyloides hyperinfection.
A 38-year-old man from Bihar, who had previously been diagnosed with lepromatous leprosy, presented to us with steroid-dependent erythema nodosum leprosum. He had been on multibacillary multidrug therapy and variable doses (20–40 mg/day) of oral prednisolone for the preceding 1 year. We initiated therapy with aspirin and thalidomide and hiked the daily dose of clofazimine to 300 mg/day while gradually tapering prednisolone. Ten weeks later, while on prednisolone 20 mg/day, he developed gradual-onset generalized weakness and edema, associated with dyspepsia, anorexia, bloating and constipation. On admission, no features of erythema nodosum leprosum were present. The patient had a cushingoid habitus, pallor, generalized pitting edema and abdominal distension. Hemogram was significant for normocytic hypochromic anemia (hemoglobin 7.2 g/dL) and peripheral eosinophilia (19%). Reticulocyte count (1%) and serum ferritin (262 μg/L) levels were within normal range. Glucose-6-phosphate dehydrogenase level, done in the previous year, was within normal limits. Serum biochemistry was unremarkable except for hypoproteinemia (total protein: 3.9 g/dL and albumin: 1.7 g/dL). Chest radiography revealed moderate right-sided pleural effusion. Ultrasonography of the abdomen showed fatty liver and moderate ascites. Repeated evaluation of stool specimens for occult blood and parasites were negative and 24-hour urinary protein levels were normal. Multibacillary multidrug therapy and treatment for erythema nodosum leprosum were continued while iron and protein supplementation along with a diuretic was initiated for anemia and anasarca. Three packed red blood cell transfusions and albumin infusions were also given. During the hospital stay, he developed an episode of dyspnea, tachypnea (24/min), tachycardia (110/min) and hypotension (90/56 mmHg) which improved with supportive therapy. During this episode, though the D-dimer levels were raised (>2000 ng/ml), venous Doppler of both legs was normal and computerized tomography-pulmonary angiogram and 2D-echocardiogram did not show features of pulmonary thromboembolism. Nonetheless, thalidomide was withdrawn. Once the patient was hemodynamically stable, an upper gastrointestinal endoscopy done to evaluate dyspepsia showed edematous duodenal mucosal folds which on histopathology revealed a large number of Strongyloides larvae within the mucosal crypts [Figure - 1]. Based on the histopathological report and retrospectively on clinical symptomatology and peripheral eosinophilia, a diagnosis of Strongyloides hyperinfection was made. Enzyme-linked immunosorbent assay for human immunodeficiency virus-1 and -2 was negative. The patient was treated with oral ivermectin 12 mg for 3 days and prednisolone was stopped to prevent disseminated infection, while aspirin and clofazimine were continued along with multibacillary multi drug therapy. There was a gradual improvement in abdominal symptoms, generalized edema, anemia and peripheral eosinophilia over 2 weeks. At discharge, the patient's hemoglobin level was 8.8 g/dL, eosinophil count was 6% and serum albumin level was 2.8 g/dL.
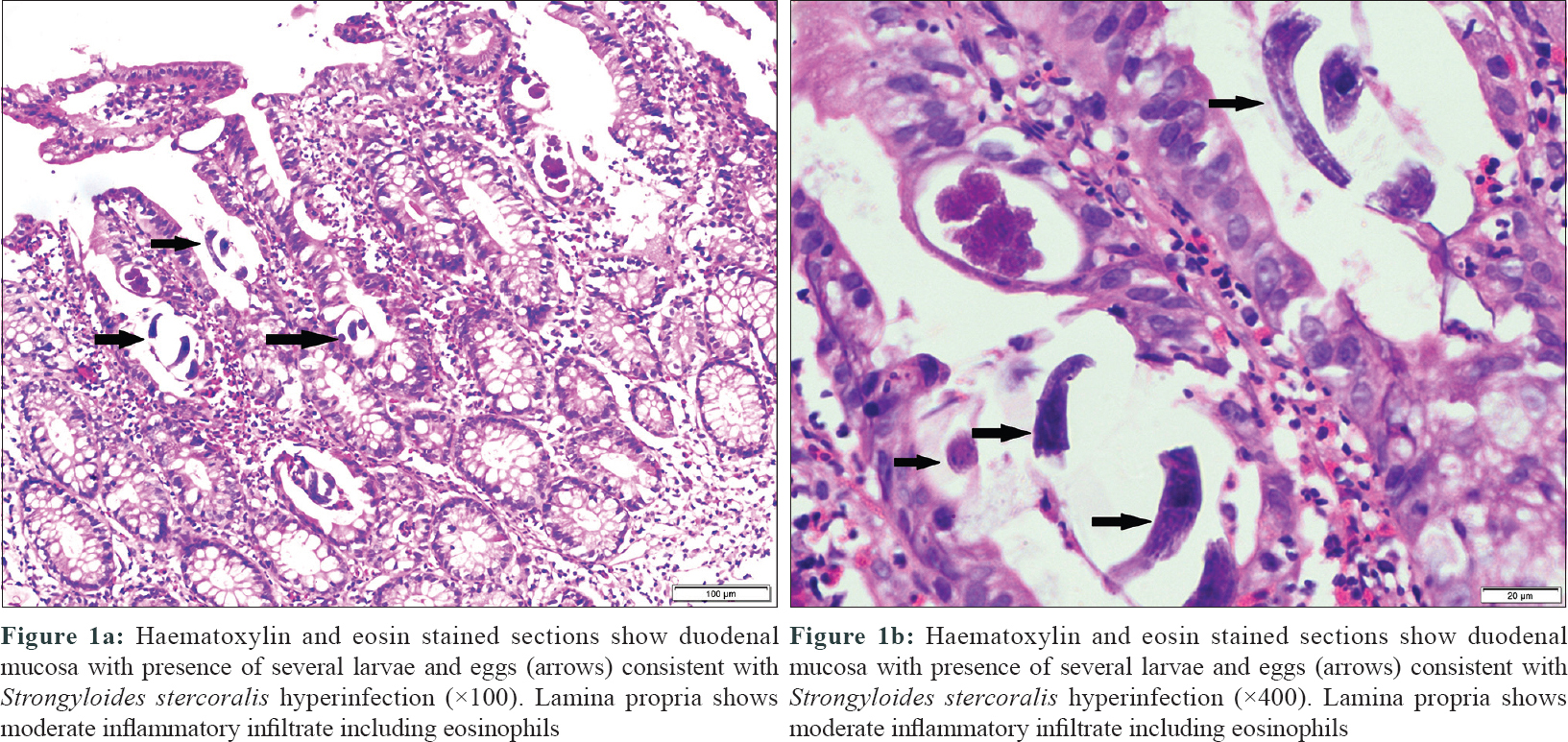 |
| Figure 1 |
S. stercoralis has an autoinfective cycle in humans which allows it to persist indefinitely in the host. Accelerated autoinfection can lead to hyperinfection in immunosuppressed individuals. Parasitologically, the difference between autoinfection and hyperinfection is just quantitative, but has not been strictly defined. Therefore, the diagnosis of hyperinfection syndrome depends on the presence of signs and symptoms attributable to an increased larval migration through the organs involved in the autoinfective cycle, namely, skin, lungs and gastrointestinal tract. Disseminated infection, on the other hand, refers to the presence of larvae in tissues other than these as well. The clinical manifestations of strongyloidosis vary widely which include fever, abdominal pain, nausea, vomiting, diarrhea or constipation, gastrointestinal bleeding, cough, hemoptysis, dyspnea and rarely, respiratory failure. Gram-negative sepsis is a dreaded complication which can prove fatal.[1] Dermatological manifestations include larva currens and purpura (the so-called “thumbprint sign” on the abdomen) and can be suggestive if present.
In our patient, the diagnosis of Strongyloides hyperinfection was initially missed, as the nonspecific gastrointestinal complaints, anemia and hypoalbuminemia were attributed to either leprosy or its treatment. Anemia and hypoalbuminemia are often seen in leprosy and erythema nodosum leprosum.[2] Dyspepsia is a well-known adverse effect of not only corticosteroids, but also nonsteroidal anti-inflammatory drugs, while both clofazimine and thalidomide can cause constipation. Strongyloides hyperinfection was an unexpected diagnosis in our case, made on the incidental detection of the parasite on duodenal biopsy. Interestingly, anemia and hypoalbuminemia are common in strongyloidiasis as well, due to bleeding from intestinal erosions and ulcers and protein-losing enteropathy, respectively.[1] In hindsight, even the unexplained episode of respiratory distress in our patient could be ascribed to the hyperinfection. Although stool examinations were repeatedly performed to exclude helminthic infestation as a cause of peripheral eosinophilia, all were negative. The sensitivity of a single-stool examination is only 30%–50% which increases with repeated examination and using stool concentration techniques. Blood agar plate culture method and serological tests are more sensitive but have limited availability.[1] Since strongyloidiasis was not suspected, we did not screen the sputum, bronchioalveolar lavage and duodenal aspirate for larvae or ova. Treatment options include ivermectin, albendazole, mebendazole and thiabendazole. Of these, ivermectin is preferred because of its better efficacy and tolerability.[1]
Diagnosis of Strongyloides hyperinfection requires a high level of clinical suspicion. It has been reported only rarely in leprosy patients, probably because the diagnosis is often overlooked owing to its nonspecific clinical features.[3],[4],[5],[6] Through this case, we wish to increase the awareness of this often fatal syndrome among dermatologists and leprologists who frequently prescribe corticosteroids for managing lepra reactions. Unexplained peripheral eosinophilia in an immunosuppressed individual having abdominal or pulmonary symptoms should raise a suspicion of strongyloidiasis.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. | Keiser PB, Nutman TB. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev 2004;17:208-17. [Google Scholar] |
| 2. | Rea TH. Decreases in mean hemoglobin and serum albumin values in erythema nodosum leprosum and lepromatous leprosy. Int J Lepr Mycobact Dis 2001;69:318-27. [Google Scholar] |
| 3. | Leang B, Lynen L, Tootill R, Griffiths S, Monchy D. Death caused by Strongyloides hyperinfection in a leprosy patient on treatment for a type II leprosy reaction. Lepr Rev 2004;75:398-403. [Google Scholar] |
| 4. | Agrawal V, Agarwal T, Ghoshal UC. Intestinal strongyloidiasis: A diagnosis frequently missed in the tropics. Trans R Soc Trop Med Hyg 2009;103:242-6. [Google Scholar] |
| 5. | De Souza JN, Machado PR, Teixeira MC, Soares NM. Recurrence of Strongyloides stercoralis infection in a patient with Hansen's disease: A case report. Lepr Rev 2014;85:58-62. [Google Scholar] |
| 6. | Mutreja D, Sivasami K, Tewari V, Nandi B, Nair GL, Patil SD. A 36-year-old man with vomiting, pain abdomen, significant weight loss, hyponatremia, and hypoglycemia. Indian J Pathol Microbiol 2015;58:500-5. [Google Scholar] |
Fulltext Views
2,198
PDF downloads
759





