Translate this page into:
Multifocal sebaceous carcinoma of the vulva
2 Department of Pathology, North Eastern Indira Gandhi Regional Institute of Health and Medical Sciences, Shillong, Meghalaya, India
Correspondence Address:
Shikha Verma
Dermatology and STD, North Eastern Indira Gandhi Regional Institute of Health and Medical Sciences, Shillong - 793 018, Meghalaya
India
| How to cite this article: Thakur BK, Verma S, Khonglah Y, Jitani A. Multifocal sebaceous carcinoma of the vulva. Indian J Dermatol Venereol Leprol 2017;83:221-224 |
Sir,
Sebaceous carcinoma is a rare skin cancer which is classified as ocular and extraocular, depending upon its location. Sebaceous carcinoma is rarely seen on the the vulva, even though it is rich in sebaceous glands.
A 55-year-old woman was referred from the urology department for asymptomatic nodulo-ulcerative lesions on the right side of the vulva for four months. She had chronic kidney disease, and was receiving antibiotics for the treatment of urinary tract infection. On physical examination, there were three ulcerated nodules, one measured 2.5 cm × 2 cm, and the other two measured 1 cm × 1.5 cm in size. They were non-tender, firm in consistency and not attached to the underlying structures [Figure - 1]. The right inguinal lymph nodes were enlarged, and hard on palpation. There was no family history of similar complaints. The laboratory investigations showed hemoglobin 5.8 gm/dl, total leukocyte count 17,000/cu.mm, blood urea 228 mg/dl and serum creatinine 4.6 mg/dl. The enzyme-linked immunosorbent assay (ELISA) for human immunodeficiency virus (HIV) was non-reactive. A skin biopsy was taken from the large nodule with a differential diagnosis of squamous cell carcinoma and cutaneous metastasis. Histopathological examination revealed an ulcerated stratified squamous epithelium and irregular lobule formation. The lobules showed sebaceous differentiation with areas of necrosis and atypical keratinized cells, which were consistent with sebaceous carcinoma [Figure - 2]. Epithelial membrane antigen and cytokeratin stains outlined clusters of sebocytes in the epidermis and dermis, with positive immunostaining of the cytoplasm in the most mature neoplastic sebocytes [Figure - 3]. A fine-needle aspiration cytology from the inguinal lymph nodes showed similar features. A whole body computed tomography scan showed no evidence of distant metastatic disease or any other malignant neoplasm. The patient was not fit for surgery due to her medical condition, and was referred to a cancer treatment center for radiotherapy.
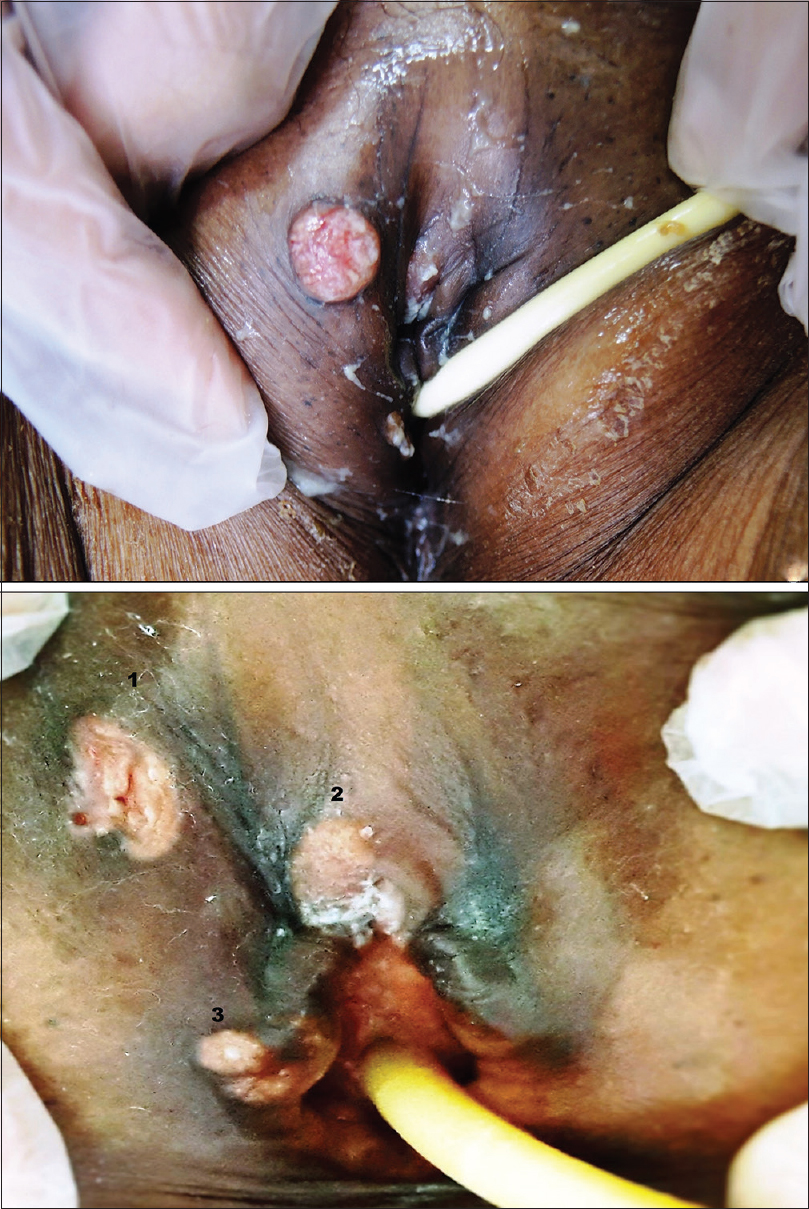 |
| Figure 1: Multiple noduloulcerative lesions on the vulva |
 |
| Figure 2 |
 |
| Figure 3 |
An early diagnosis of sebaceous carcinoma is essential due to its aggressive nature and metastatic potential. These neoplasms have a 30–40% risk of local tumor recurrence, a 20–25% risk of distant metastases and a 10–20% risk of tumor-related mortality.[1] In previous reports, vulval sebaceous carcinoma was located on the labia majora in five cases, and on the labia minora in three cases. It occurred in the age group of 31–89 years. Metastasis was present at the time of diagnosis in three cases, one of which was in the lung, and the other two were in the lymph nodes.[2],[3],[4] The details of previously reported cases are shown in [Table - 1]. One peculiar finding in our case is that there were three lesions, one on the labia majora, and two on the labia minora. The reason for multiple sebaceous carcinomas in our patient could be immunosuppression, induced by the uremic milieu of kidney failure. Uremia and retention of metabolic waste cause intestinal barrier dysfunction and endotoxemia. Decreased protein catabolism causes increased complement turnover, and extensive proteinuria results in a loss of proteins such as immunoglobulin and zinc binding protein, which affect immune function.[10]

Sebaceous carcinoma is usually associated with a genetic predisposition to Muir–Torre syndrome. This is an autosomal dominant disease, characterized by the association of a skin tumor of sebaceous lineage with or without a keratoacanthoma. It is also associated with one or more visceral neoplasms.[11] The diagnosis of Muir–Torre syndrome seems unlikely in our patient as no other malignancies were detected. Bowen's disease has been reported in two patients with sebaceous carcinoma of vulva.[5],[6] However, human papillomavirus infection has not been documented in genital sebaceous carcinoma. Patients with sebaceous carcinoma and inguinal lymph node metastases have a poor prognosis and regional radiotherapy is recommended for such patients.[7] Hemivulvectomy was the most common surgery performed in previous reports. Sentinel node biopsy should be used in conjunction with wide local excision of the primary tumor to investigate the possibility of subclinical regional metastases.[7]
We were able to find only eight previous reports of vulval sebaceous carcinoma and none of multifocal disease. The risk factors predisposing to multiple carcinomas should be properly evaluated and managed. Additional case series with their outcome will help establish the natural course and optimum management of this rare neoplasm.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Moreno C, Jacyk WK, Judd MJ, Requena L. Highly aggressive extraocular sebaceous carcinoma. Am J Dermatopathol 2001;23:450-5.
[Google Scholar]
|
| 2. |
Ikuse S, Jinbou A, Matsushima I. A case of sebaceous carcinoma. Jpn J Dermatol 1976;86:783.
[Google Scholar]
|
| 3. |
Kawamoto M, Fukuda Y, Kamoi S, Sugisaki Y, Yamanaka N. Sebaceous carcinoma of the vulva. Pathol Int 1995;45:767-73.
[Google Scholar]
|
| 4. |
Khan Z, Misra G, Fiander AN, Dallimore NS. Sebaceous carcinoma of the vulva. BJOG 2003;110:227-8.
[Google Scholar]
|
| 5. |
Jacobs DM, Sandles LG, Leboit PE. Sebaceous carcinoma arising from Bowen's disease of the vulva. Arch Dermatol 1986;122:1191-3.
[Google Scholar]
|
| 6. |
Escalonilla P, Grilli R, Cañamero M, Soriano ML, Fariña MC, Manzarbeitia F, et al. Sebaceous carcinoma of the vulva. Am J Dermatopathol 1999;21:468-72.
[Google Scholar]
|
| 7. |
Pusiol T, Morichetti D, Zorzi MG. Sebaceous carcinoma of the vulva: Critical approach to grading and review of the literature. Pathologica 2011;103:64-7.
[Google Scholar]
|
| 8. |
Rulon DB, Helwig EB. Cutaneous sebaceous neoplasms. Cancer 1974;33:82-102.
[Google Scholar]
|
| 9. |
Carlson JW, McGlennen RC, Gomez R, Longbella C, Carter J, Carson LF. Sebaceous carcinoma of the vulva: A case report and review of the literature. Gynecol Oncol 1996;60:489-91.
[Google Scholar]
|
| 10. |
Kurts C, Panzer U, Anders HJ, Rees AJ. The immune system and kidney disease: Basic concepts and clinical implications. Nat Rev Immunol 2013;13:738-53.
[Google Scholar]
|
| 11. |
Chao AN, Shields CL, Krema H, Shields JA. Outcome of patients with periocular sebaceous gland carcinoma with and without conjunctival intraepithelial invasion. Ophthalmology 2001;108:1877-83.
[Google Scholar]
|
Fulltext Views
2,208
PDF downloads
1,045





