Translate this page into:
Viability of Mycobacterium leprae in the environment and its role in leprosy dissemination
2 Department of Biotechnology, Aligarh Muslim University, Aligarh, Uttar Pradesh, India
3 Department of Microbiology and Molecular Biology, Model Rural Health Research Unit, Ghatampur, Uttar Pradesh, India
Correspondence Address:
Partha Sarathi Mohanty
National JALMA Institute for Leprosy and Other Mycobacterial Diseases, Dr. M Miyazaki Marg, Tajganj, Agra - 282 004, Uttar Pradesh
India
| How to cite this article: Mohanty PS, Naaz F, Katara D, Misba L, Kumar D, Dwivedi DK, Tiwari AK, Chauhan DS, Bansal AK, Tripathy SP, Katoch K. Viability of Mycobacterium leprae in the environment and its role in leprosy dissemination. Indian J Dermatol Venereol Leprol 2016;82:23-27 |
Abstract
Background: Leprosy, a chronic disease caused by Mycobacterium leprae, is a public health concern in certain countries, including India. Although the prevalence of the disease has fallen drastically over time, new cases continue to occur at nearly the same rate in many regions. Several endemic pockets have been observed in India and elsewhere. The precise dynamics of leprosy transmission are still not clearly understood. Both live bacilli as well as M. leprae DNA have been detected in the soil and water of endemic areas; they possibly play an important role in disease transmission. Aims: To study the occurrence of viable M. leprae in environmental samples collected from areas of residence of patients with active leprosy. Methods: The study was conducted on 169 newly diagnosed leprosy patients in Ghatampur, Uttar Pradesh, India. Soil and water samples were collected from their areas of residence using a standardized protocol. An equal number of soil and water samples were also collected from non-patient areas of the same or adjoining villages. The environmental samples collected from the patients surroundings were subjected to 16S ribosomal RNA gene analysis after obtaining informed consent. Results: About a quarter of the environmental samples collected from patient areas, (25.4% of soil samples and 24.2% of water samples) were found to be positive for specific 16S ribosomal RNA genes of M. leprae. Environmental samples collected from non-patient areas were all found negative for M. leprae 16S ribosomal RNA genes. Limitations: The major limitation of the study was that the sample size was small. Conclusion: The study demonstrated the presence of viable strains of M. leprae in skin smear samples of paucibacillary patients and multibacillary patients, as well as in the environmental samples obtained from around their houses. This could play an important role in the continued transmission of leprosy.Introduction
Leprosy is a slow and chronic infection caused by Mycobacterium leprae, affecting both sexes and all age groups, in many parts of the world. According to the WHO report, leprosy is a public health problem in 105 countries (including 28 in Africa, 28 in the Americas, 11 in South East Asia, 22 in the Eastern Mediterranean region and 16 in the Western Pacific region).[1] India achieved 'elimination' (prevalence < 1/10000) in December 2005 but new cases of leprosy continue to be detected in some endemic pockets. A total of 542 (84.7%) districts out of total 640 districts also achieved 'elimination' of leprosy by March 2012.[2] However, the new case detection rate continues to pose a challenge for program managers as it has either remained stationary or showed only a very slow decline since 2007.
Numerous studies suggest that leprosy is transmitted from person to person by close contact of a healthy individual with an infectious patient. Till date, the exact mechanisms of leprosy transmission are not clearly understood. Even the widely advocated methods of spread including person to person contact or contact with respiratory secretions from infected individuals have not been conclusively established so far. Different authors have suggested that M. leprae may be present in the soil, in water, on plants or in various animal species including amoeba, insects, fish, primates and armadillos.[3],[4],[5],[6],[7],[8],[9],[10],[11],[12],[13],[14],[15] The role of soil and water in the transmission of leprosy has only been speculated upon; it is yet to be recognized and supported by experimental proof. Indirect evidence for this possibility comes from the fact that in the recent past, contaminated water supply systems have been responsible for several hospital and community outbreaks of mycobacterial infections.[16]
Studying transmission dynamics is complicated by the inability to grow M. leprae in culture and the lack of animal models. Infection from the animal source, that is, armadillos (Dasypus novemcinctus) has been ruled out in India since armadillos are not found in nature in this country. The nearly constant detection of new cases in some pockets, with no clear cut history of close and prolonged contact with leprosy patients, indicates that other reservoirs of infection, possibly environmental, could be responsible for the continued transmission of leprosy.
Methods
Collection of environmental samples
Soil and water samples were collected from different places of Ghatampur, Kanpur Nagar, Uttar Pradesh, an endemic pocket for leprosy. The environmental samples (both soil and water) were collected from 169 patient areas and 169 non-patient areas. Patient areas included the vicinity of houses where patients lived and dwelled. Different portions of the same or adjoining villages, where leprosy cases had not been detected during the last 5–6 years, were designated as non-patient areas. To collect soil samples, soil was dug (4”deep) and collected in clean plastic containers (10 g each) with the help of a “khurpi” and labelled with the patient ID and the village name. Likewise, water samples were collected from the drainage outlet of the patient's (or non-patient's) house, bathing place and drinking water source (bore well). The collected samples were transported to the research centre (National JALMA Institute for Leprosy and Other Mycobacterial Diseases, Tajganj, Agra, UP, India) and stored at 4–8°C till they were processed further. The geomapping of patient samples was done using Geoplaner online software (www. geoplaner.com).
Isolation of Mycobacterium leprae total RNA
Extraction of M. leprae total RNA from soil and water samples was done by the method of Miskin et al.[17] Briefly, 5 ml (1 mg/ml) of soil suspension was taken in a sterile 25 ml screw-capped tube with sterile glass beads and centrifuged at 8000 g for 30 minutes. The pellets were treated with 2 ml of extraction buffer (0.12M sodium phosphate buffer, 10 mg lysozyme, 1% β-mercaptoethanol) and shaken for 20 seconds. The homogenates were transferred to new vials containing 500 µl of 10% (w/v) sodium dodecyl sulfate, incubated at 80°C for 30 minutes with vigorous shaking at 10 minute intervals in a water bath and centrifuged at 2800 g for 15 minutes at 4°C. The supernatants were transferred to fresh tubes and held on ice while the pellets were re-extracted with 2.5 ml of extraction buffer and re-centrifuged at 2800 g for 15 minutes at 4°C. The supernatants were pooled, mixed with twice their volume of 30% polyethylene glycol 6000 and kept for 2 hours at room temperature for nucleic acid precipitation and centrifuged at 5000 g for 30 minutes at 4°C. The pellets were re-suspended in 1 ml diethylpyrocarbonate treated water (to inactivate RNase enzymes). One hundred microliters of 7.5 M potassium acetate were added to make a final concentration of 0.5 M and it was centrifuged at 8000 g for 5 minutes to remove the precipitated humic acid (which interferes with polymerase chain reactions). Nucleic acid was precipitated with the addition of twice the volume of chilled ethanol and keeping overnight at −20°C. RNA was pelleted by centrifuging at 8000 g for 15 minutes and dissolved in 20 µl of diethylpyrocarbonate-treated/HPLC-grade water. RNA preparations were treated with DNaseI for removing any traces of contaminating DNA. DNaseI was inactivated by incubation at 80°C for 10 min. Then isolated RNA was stored at −70°C till further use.
Preparation of complementary DNA and polymerase chain reaction amplification of 16S ribosomal RNA gene
Complementary DNA synthesis was done using Revert AidFirst strand complementary DNA synthesis kit (Fermentas) from the RNA of environmental samples. Prepared complementary DNA was stored at −20°C for further use. The 16S ribosomal RNA gene of M. leprae was amplified using the primers F-5' TCGAACGGAAAGGTCTCTAAAAAATC 3' and R-5' CCTGCACCGCAAAAAGCTTTCC 3' to determine the presence of viable M. leprae in environmental samples (soil and drainage water samples).[18]
Sequencing of 16S ribosomal RNA gene
Polymerase chain reaction products of the 16S ribosomal RNA gene were purified using polymerase chain reaction clean-up kit. All the polymerase chain reaction products were sequenced directly in the ABI 3031XL big dye terminator sequencer according to the manufacturer's instructions.
Basic local alignment search tool (BLAST) search and sequence alignment
Initially, the sequences were subjected to the basic local alignment search tool search (BLAST) at NCBI to determine their molecular taxonomic identity. For basic local alignment search tool search, sequences were converted to FASTA format and entered into the NCBI web page (http://blast.ncbi.nlm.nih.gov/blast), selecting the reference data domain as nucleotide collection (nt/nr) for highly similar megablast search. The taxonomic identities of the strains were determined after comparing the search results. Five sequences from basic local alignment search tool search results were aligned with our sequences. The sequence alignment was done using MEGA 4.0 (www.megasoftware.net.mega4).
Results
Of the patients from whom 169 slit skin smear samples were collected, 4 were indeterminate, 93 were borderline tuberculoid (BT) type, 66 were (BB) type, 5 were borderline lepromatous (BL) type and 1 was found to be lepromatous (LL) type diagnosed clinically. These 169 patients belonged to 132 nuclear and joint families hailing from 96 villages. The mapping of patient samples is depicted in [Figure - 1]. Of the environmental samples collected from the areas where patients were residing, 43 (25.4%) soil samples and 41 (24.2%) water samples were found to be positive for 16S ribosomal RNA on amplifying the product of complementary DNA. In 37 (21.8%) areas, both soil and water samples were positive for 16S ribosomal RNA. The environmental samples showing positivity were obtained from the areas where 34 multibacillary and 17 paucibacillary patients resided. The amplified product of the 16S ribosomal RNA gene was about 172 bp [Figure - 2]. On the other hand, no environmental samples taken from non-patient areas were found positive for M.leprae 16S ribosomal RNA. Basic local alignment search tool (BLAST) search results confirmed that the sequenced product was the 16S ribosomal RNA gene of M. leprae. The cDNA sequences from soil and water samples clearly matched with M. leprae Tamil Nadu strain (AL583920), M. leprae cosmid B1549 (U00014), M. leprae strain (X55022), Mycobacterium lepromatosis Br1 (GQ900374) and Uncultured bacterium clone (JQ374245), with identities scores of 100%, 100%, 100%, 97% and 96%, respectively. The nucleotide differences between M. leprae and uncultured bacteria were found in the nucleotide positions 1276 (G to A), 1315 (T to C), 1316 (G to A), 1342 (G to A), 1355 (A to T) and differences between M. leprae and M. lepromatosis were in the nucleotide positions 1315 (T to C), 1378 (A to G), 1387 (A to G) and 1409 (C to T) [Figure - 3].
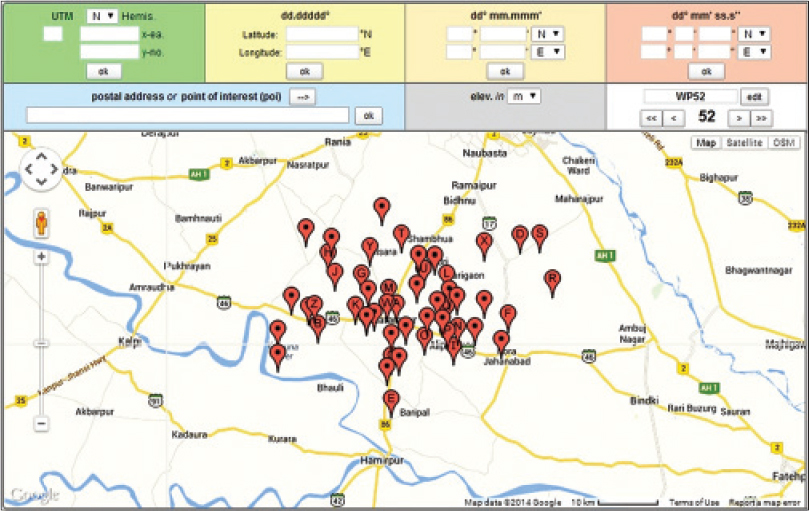 |
| Figure 1: Geographic location and the prevalence of leprosy in environmental samples collected from Ghatampur tehsil. The spots showed the samples collected from the patient area |
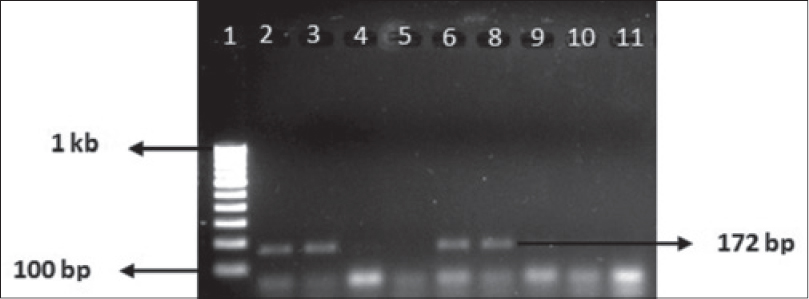 |
| Figure 2: Polymerase Chain Reaction product of 16S ribosomal RNA gene amplified from complementary DNA. Lane 1: 100 bp ladder, Lane 2: Positive control, Lane 3–10: Environmental samples, Lane 11: Negative control |
 |
| Figure 3: Multiple sequence alignment of partial sequence 16S ribosomal RNA gene of Mycobacterium leprae, Mycobacterium leprae cosmid, Mycobacterium lepromatosis and uncultured bacterium |
Discussion
Leprosy is still a public health problem in several countries including India. The disease is said to be transmitted from person to person by respiratory secretions from infected individuals but this theory has not been conclusively proven so far. Several studies have provided insight into the non-human sources of M. leprae.[5], 7, [19],[20],[21] There is growing recognition in recent years that soil and water may be important vehicles of transmission of M. leprae and could be responsible for the continued presence of leprosy. In the soil, several microorganisms live in the same niche in a symbiotic or commensal relationship. M. leprae is known to survive in the soil for upto 40 days.[22] Our study investigated the soil and water obtained from patient residential areas as well as non-patient areas; we had a larger sample size compared to that of earlier studies from other parts of India.[3],[20],[21] Using the specific primers designed for M. leprae by Jadhav et al. we specifically amplified the 172 bp 16S ribosomal RNA gene of M. leprae.[18] In this study, RNA was used instead of DNA to detect M. leprae not only for its presence but also to find out the viability of M. leprae in the soil. We found that 25.6% soil and 24.2% water samples from patient areas were positive for viable leprosy bacilli. This might have ome about due to the movement of an infected person in the surrounding environment and resultant shedding of leprosy bacilli, which then continue to live in the environment by symbiotic or commensal mechanisms Our results corroborate with the work of different researchers who have provided evidence of the existence of viable M. leprae in environmental samples taken from areas where patients reside.[3],[20],[21] Those environmental samples that we collected from the non-patient areas did not show the presence of M. leprae. Thus, the environmental M. leprae is a connective link in the continuation of leprosy in several endemic pockets where multiple cases are detected in the same family or in nearby families. The GenBank accession number JQ374245 represents an uncultured bacterium found in 6 terrestrial ecosystems of USA, having 96% similarity with the 16S ribosomal RNA gene of M. leprae.[23] As the bacterium had 96% similarity with M. leprae and was unculturable, we conclude that the bacterium found in the terrestrial ecosystem might be M. leprae. Matsuoaka et al.[5] strongly suggested that non-human sources such as soil and water may be responsible for continued leprosy transmission. Jadin reported the presence of M. leprae in amoeba, thus confirming our results in favor of the existence of M. leprae in the environment and its role in further transmission.[24]
There are two limitations of this study (1) the sample size of the study was very small (2) 16S rRNA gene analysis for viability study was limited to environmental samples only.
Conclusion
Our study showed the presence of viable M. leprae in the soil and water of the patient areas. To confirm the role of environmental M. leprae in leprosy transmission, more extensive studies are required.
Acknowledgment
The study was financially supported by Indian Council of Medical Research. We are highly grateful to Dr. S.R. Rathore, Shailendra Chauhan, Mohd Wasim, Dinesh Singh, Atul Saraswat, Pradip Mishra, Ashok Tiwari, Jitendra Chaurasia, Narendra Singh, Mahendra Singh, Shivkaran, Ram Singh, Nause, Pawan and Raju for their constant support in the field at the time of sample collection and transportation. We are highly grateful to Dr. VM Katoch, Former Secy DHR and DG ICMR for his continuous guidance in the improvement of this study.
Financial support and sponsorship
Indian Council of Medical Research, Ansari Nagar, New Delhi.
Conflicts of interest
The authors declare no conflicts of interest.
| 1. |
Global leprosy situation. Wkly Epidemiol Rec 2012;87:317-28.
[Google Scholar]
|
| 2. |
National Leprosy Eradication Programme. NLEP-Progress Report for the Year 2012-2013. Central Leprosy Division. New Delhi, India; 2013.
[Google Scholar]
|
| 3. |
Lavania M, Katoch K, Katoch VM, Gupta AK, Chauhan DS, Sharma R, et al. Detection of viable Mycobacterium leprae in soil samples: Insights into possible sources of transmission of leprosy. Infect Genet Evol 2008;8:627-31.
[Google Scholar]
|
| 4. |
Blake LA, West BC, Lary CH, Todd JR 4th. Environmental nonhuman sources of leprosy. Rev Infect Dis 1987;9:562-77.
[Google Scholar]
|
| 5. |
Matsuoka M, Izumi S, Budiawan T, Nakata N, Saeki K. Mycobacterium leprae DNA in daily using water as a possible source of leprosy infection. Indian J Lepr 1999;71:61-7.
[Google Scholar]
|
| 6. |
Kazda J, Irgens LM, Müller K. Isolation of non-cultivable acid-fast bacilli in sphagnum and moss vegetation by foot pad technique in mice. Int J Lepr Other Mycobact Dis 1980;48:1-6.
[Google Scholar]
|
| 7. |
Kazda J, Ganapati R, Revankar C, Buchanan TM, Young DB, Irgens LM. Isolation of environment-derived Mycobacterium leprae from soil in Bombay. Lepr Rev 1986;57 Suppl 3:201-8.
[Google Scholar]
|
| 8. |
Lahiri R, Krahenbuhl JL. The role of free-living pathogenic amoeba in the transmission of leprosy: A proof of principle. Lepr Rev 2008;79:401-9.
[Google Scholar]
|
| 9. |
Saha K, Jain M, Mukherjee MK, Chawla NM, Chaudhary DS, Prakash N. Viability of Mycobacterium leprae within the gut of Aedes aegypti after they feed on multibacillary lepromatous patients: A study by fluorescent and electron microscopes. Lepr Rev 1985;56:279-90.
[Google Scholar]
|
| 10. |
Sreevatsa. Leprosy and arthropods. Indian J Lepr 1993;65:189-200.
[Google Scholar]
|
| 11. |
Hutchinson J. A statement of facts and explanations. On Leprosy and Fish-Eating. London: Archibald Constable and Co.; 1906.
[Google Scholar]
|
| 12. |
Chaussinand R. Inoculation of Hansen and stefansky bacilli in the rainbow perch Eupomotis gibbosus. Preliminary note (Abstract). Int J Lepr Other Mycobact Dis 1952;20:420-1.
[Google Scholar]
|
| 13. |
Couret M. The behavior of bacillus leprae in cold-blooded animals. J Exp Med 1911;13:576-89.
[Google Scholar]
|
| 14. |
Gormus BJ, Xu K, Baskin GB, Martin LN, Bohm RP Jr, Blanchard JL, et al. Experimental leprosy in rhesus monkeys: Transmission, susceptibility, clinical and immunological findings. Lepr Rev 1998;69:235-45.
[Google Scholar]
|
| 15. |
Truman RW, Shannon EJ, Hagstad HV, Hugh-Jones ME, Wolff A, Hastings RC. Evaluation of the origin of Mycobacterium leprae infections in the wild armadillo, Dasypus novemcinctus. Am J Trop Med Hyg 1986;35:588-93.
[Google Scholar]
|
| 16. |
Conger NG, O'Connell RJ, Laurel VL, Olivier KN, Graviss EA, Williams-Bouyer N, et al. Mycobacterium simaeoutbreak associated with a hospital water supply. Infect Control Hosp Epidemiol 2004;25:1050-5.
[Google Scholar]
|
| 17. |
Miskin IP, Farrimond P, Head IM. Identification of novel bacterial lineages as active members of microbial populations in a freshwater sediment using a rapid RNA extraction procedure and RT-PCR. Microbiology 1999;145(Pt 8):1977-87.
[Google Scholar]
|
| 18. |
Jadhav RS, Kamble RR, Shinde VS, Edward S, Edward VK. Use of reverse transcription polymerase chain reaction for the detection of Mycobacterium leprae in the slit-skin smears of leprosy patients. Indian J Lepr 2005;77:116-27.
[Google Scholar]
|
| 19. |
Desikan KV. Viability of Mycobacterium leprae outside the human body. Lepr Rev 1977;48:231-5.
[Google Scholar]
|
| 20. |
Lavania M, Katoch K, Sachan P, Dubey A, Kapoor S, Kashyap M, et al. Detection of Mycobacterium leprae DNA from soil samples by PCR targeting RLEP sequences. J Commun Dis 2006;38:269-73.
[Google Scholar]
|
| 21. |
Turankar RP, Lavania M, Singh M, Siva Sai KS, Jadhav RS. Dynamics of Mycobacterium leprae transmission in environmental context: Deciphering the role of environment as a potential reservoir. Infect Genet Evol 2012;12:121-6.
[Google Scholar]
|
| 22. |
Chakrabarty AN, Dastidar SG. Is soil an alternative source of leprosy infection? Acta Leprol 2001-2002;12:79-84.
[Google Scholar]
|
| 23. |
Dunbar J, Eichorst SA, Gallegos-Graves LV, Silva S, Xie G, Hengartner NW, et al. Common bacterial responses in six ecosystems exposed to 10 years of elevated atmospheric carbon dioxide. Environ Microbiol 2012;14:1145-58.
[Google Scholar]
|
| 24. |
Jadin JB. Amibes limax vecteurs possible de Mycobacteries et de M. leprae. Acta Leprol 1975;59:57-67.
[Google Scholar]
|
Fulltext Views
5,122
PDF downloads
1,979





