Translate this page into:
Treatment of multifocal epithelial hyperplasia with imiquimod
Correspondence Address:
Nikoo Mozafari
Skin Research Center, Shohada-e Tajrish Hospital, Tajrish Square, Tehran
Iran
| How to cite this article: Barikbin B, Tehranchinia Z, Mozafari N. Treatment of multifocal epithelial hyperplasia with imiquimod. Indian J Dermatol Venereol Leprol 2014;80:175-177 |
Sir,
Multifocal epithelial hyperplasia or Heck′s disease is a rare benign disorder, characterized by small, soft, pink or white, slightly elevated papules on the buccal, gingival or labial mucosa. [1] It is an uncommon asymptomatic proliferation of the oral mucosa due to human papillomavirus (HPV) 13 or 32 infection. [1] The lesions are often asymptomatic and show little propensity for spontaneous resolution. They can be irritated by accidental biting and can interfere with occlusion and mastication. However, most patients request treatment for aesthetic reasons. [1] Treatment is difficult. Destructive or ablative modalities such as electrocautery, cryosurgery, surgery, CO 2 laser vaporization and trichloroacetic acid (TCA) are effective, but are associated with high recurrence rates. [1]
Recently, imiquimod 5% cream has been reported to successfully control lesions associated with Heck′s disease. [2],[3],[4] According to these reports, no recurrences have been observed after a follow-up period of up to 12 months. [2],[3],[4] We report a case of multifocal epithelial hyperplasia, successfully treated with topical imiquimod 5% cream. Despite evidence of partial recurrence after discontinuation of treatment, the final results were acceptable.
A 19-year-old girl was referred to our dermatology clinic with an over 10-year history of oral lesions that gradually increased in size and number. Family history revealed similar oral lesions affecting her 13-year-old brother. Physical examination revealed multiple, firm mucosa colored papules situated on the buccal mucosa bilaterally and on the lower labial mucosa [Figure - 1]. The lesions ranged in size from 2 mm to 10 mm in diameter and many of them had coalesced giving a cauliflower-like appearance. Histological examination revealed acanthosis, widening of rete ridges and koilocytic changes within the epithelium indicating HPV infection [Figure - 2]. Polymerase chain reaction (PCR) confirmed the presence of HPV and it was positive for subtype 13. The clinical and histopathological features were consistent with multifocal epithelial hyperplasia.
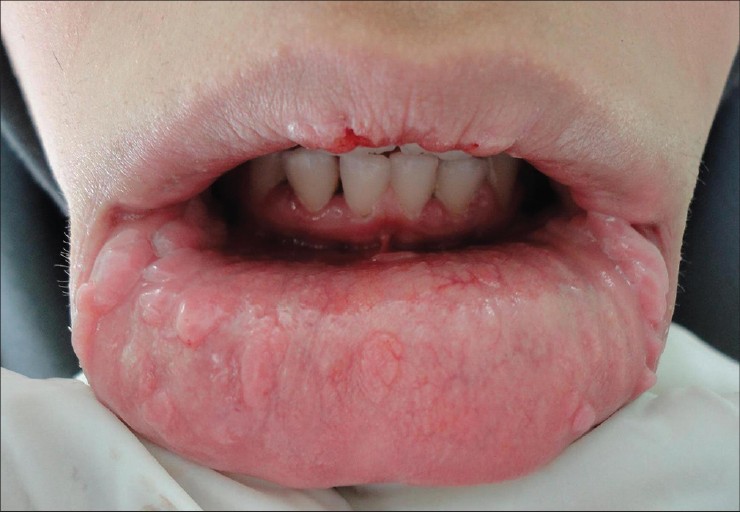 |
| Figure 1: Multiple flat and coalescent papules and nodules are seen on the lip mucosa |
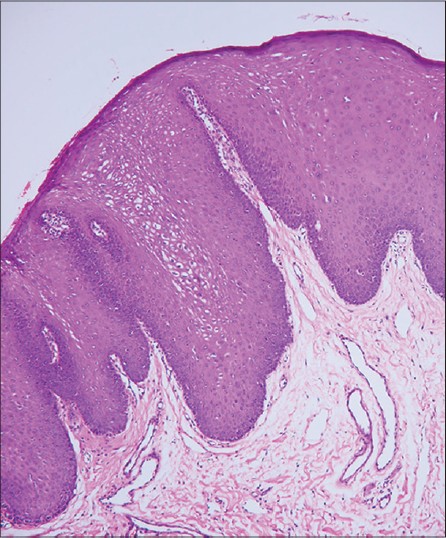 |
| Figure 2: Acanthotic epithelium with elongated and broad rete ridges vacuolated keratinocytes with pyknotic nuclei are seen (H and E, ×10) |
Cryotherapy was applied at 2 weeks intervals but no significant reduction of lesions was observed after six sessions. No improvement was noted after four sessions of weekly application of TCA 80%. At this point, the patient accepted a trial of imiquimod therapy (Aldara 5% cream 3M pharmaceuticals, UK). There is no recommendation on optimal frequency and duration of imiquimod treatment for multifocal epithelial hyperplasia. Based on the recommended regimen for the treatment of external genital warts and on previous studies of the treatment of multifocal epithelial hyperplasia with imiquimod, the patient was instructed to apply the cream on the oral lesions overnight for at least 8 h and to wash the area with water at the end of this period, once every other night. Improvement started 4 weeks after beginning treatment. After 8 weeks of therapy, local reactions including erythema and erosions appeared on the sites of application which resolved by temporary discontinuation of treatment for 1 week. Thereafter, treatment intervals were extended to every 2 days. Significant reduction in lesion size occurred at the 12 th week and therapy was continued for 16 weeks until complete resolution [Figure - 3]. During the last month of therapy, the patient complained of decreased appetite and weight loss. She reported hair loss, intolerable fatigue and muscle weakness.
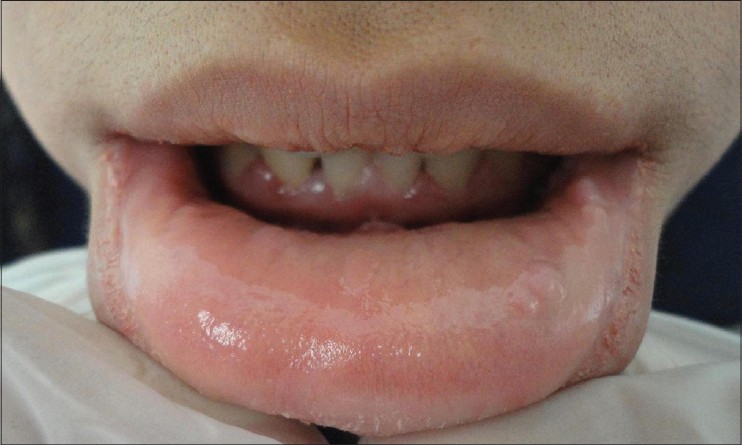 |
| Figure 3: Complete resolution of the lesions at the end of treatment |
Recurrence of lesions was noted at 6 months of follow-up [Figure - 4]. Lesions were smaller in size than at presentation and did not appear larger at 1 year of follow-up. The patient was satisfied with the final outcome and refused to consider another course of therapy.
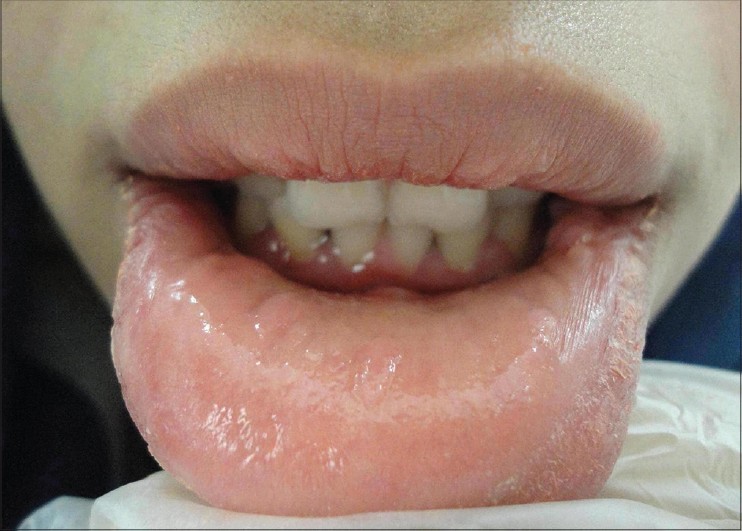 |
| Figure 4: Partial recurrence of the lesions |
Imiquimod is a toll-like receptor agonist that activates antigen presenting cells and enhances production of interferon alpha (INF-α) and INF-γ which stimulate cytotoxic T lymphocytes to kill virally infected cells leading to wart regression and normalization of keratinocyte proliferation. [5] Imiquimod was first reported for the treatment of multifocal epithelial hyperplasia in a 9-year-old boy in whom lesions disappeared after 3 months of treatment. In that case, imiquimod was used 3 times a week and no recurrence was observed at follow-up 5 months later. [3] Subsequently, five other cases of multifocal epithelial hyperplasia resistant to cryotherapy or TCA 80% and successfully treated with imiquimod were reported. [2],[4] Lesions in all paitents disappeared after 2-4 months of treatment. There was no recurrence after up to 1 year of follow-up. Except minor local discomfort, no serious systemic side-effects were reported. [2],[4] Herpes labialis in two patients, and mild lower back pain and headache in one patient were the only side-effects observed. [2] Local inflammatory reaction following imiquimod treatment is due to release of endogenous cytokines. It is closely associated with the efficacy of imiquimod treatment and therefore is seen in almost all patients responding to imiquimod. [6]
We were concerned about intraoral application of imiquimod because of the possibility of greater systemic drug absorption through mucosa and a greater risk of systemic side-effects. There is no published pharmacokinetic data on mucosal application of imiquimod and in general, oral application of imiqumod is prohibited (Aldara patient information leaflet). However, intraoral application of imiquimod has been successfully used for oral leukoplakia and intraoral melanoma with no systemic side-effects. [7] In previously reported cases of Heck′s disease and in our case, imiquimod showed an acceptable safety profile. [2],[3],[4]
Overall, topical imiquimod appears to be a novel, and effective treatment for oral multifocal epithelial hyperplasia that is also non-invasive. However, controlled administration of imiquimod cream for oral lesions is difficult. Indeed, unpleasant taste and texture of the drug in the mouth reduces patient compliance and the presence of saliva and swallowing washes away most of the drug from the site of application resulting in low therapeutic effects. Development of a new formulation for topical drug delivery within the oral cavity would be helpful.
Acknowledgment
We are grateful to Dr. Alireza Nadji, Virology Department of Shahid Beheshti University of Medical Sciences, for his assistance in the detection and typing of HPV by PCR technique.
| 1. |
Said AK, Leao JC, Fedele S, Porter SR. Focal epithelial hyperplasia-an update. J Oral Pathol Med 2013;42:435-42.
[Google Scholar]
|
| 2. |
Yasar S, Mansur AT, Serdar ZA, Goktay F, Aslan C. Treatment of focal epithelial hyperplasia with topical imiquimod: Report of three cases. Pediatr Dermatol 2009;26:465-8.
[Google Scholar]
|
| 3. |
Maschke J, Brauns TC, Goos M. Imiquimod for the topical treatment of focal epithelial hyperplasia (Heck disease) in a child. J Dtsch Dermatol Ges 2004;2:848-50.
[Google Scholar]
|
| 4. |
Ponte P, Serrão V, Fiadeiro T. Focal epithelial hyperplasia (Heck's Disease) in two siblings: Response to treatment with imiquimod 5% cream. Eur J Dermatol 2010;20:248-9.
[Google Scholar]
|
| 5. |
Jackson JM, Callen JP. Immunomodulators. In: Bolognia JL, Jorizzo JL, Schaffer JV, editors. Dermatology, 3 rd ed. Beijing: Elsevier Saunders; 2012. p. 2134.
[Google Scholar]
|
| 6. |
Schöfer H. Evaluation of imiquimod for the therapy of external genital and anal warts in comparison with destructive therapies. Br J Dermatol 2007;157 Suppl 2:52-5.
[Google Scholar]
|
| 7. |
Allam JP, Erdsach T, Wenghoefer M, Bieber T, Appel TR, Novak N. Successful treatment of extensive human papillomavirus-associated oral leucoplakia with imiquimod. Br J Dermatol 2008;158:644-6.
[Google Scholar]
|
Fulltext Views
7,259
PDF downloads
1,302





