Translate this page into:
Directly observed treatment short course and cutaneous tuberculosis: Our experience
Correspondence Address:
G Raghu Rama Rao
Consultant Dermatologist, Surya Skin Care and Research Center, #15-1-2, Gopal Sadan, Naoroji Road, Maharanipeta, Visakhapatnam - 530 002, Andhra Pradesh
India
| How to cite this article: Rama Rao G R, S, Narayan BL, Amareswar A, Sandhya S. Directly observed treatment short course and cutaneous tuberculosis: Our experience. Indian J Dermatol Venereol Leprol 2011;77:330-332 |
Sir,
Currently, the treatment of all types of tuberculosis is by the directly observed treatment short course (DOTS) strategy, which is implemented by World Health Organization (WHO). [1] This programme has been fully adapted by the Revised National Tuberculosis Control Programme (RNTCP) of the Government of India. [2] Based on the nature and severity of the disease, tuberculosis patients are grouped into three categories for the purpose of the DOTS administration. DOTS has two phases - 2 months of intensive phase and 4 months of continuation phase, and, in severe cases, the treatment is extended to 8-12 months. Most DOTS regimens have thrice-weekly schedules (Monday-Wednesday-Friday). For cutaneous tuberculosis, category III (2H 3 R 3 Z 3 + 4H 3 R 3 ) is recommended. In this category, Rifampicin (R-450 mg), Isoniazid (H-600 mg) and Pyrazinamide (Z-1500 mg) are administered for three days in a week for 2 months (intensive phase), followed by R-450 mg and H-600 mg given three days in a week as a continuation phase for 4 months. In case of cutaneous tuberculosis with systemic infections, or when more than one group of lymph nodes are involved, category I with four drugs is advised. Depending on the clinical response, the duration of treatment can be extended under the guidance of the clinician. For children and adults who weigh less than 30 kg, these drugs are administered according to their weight.
Although the DOTS strategy has been in operation for more than 10 years and has been found to be effective, barring a few reports, [3],[4],[5],[6] there are no specific studies on DOTS in the management of cutaneous tuberculosis. Hence, this study has been designed to evaluate the efficacy, tolerability and safety profile of DOTS in cutaneous tuberculosis and also to know how soon cutaneous tuberculosis responds to DOTS.
During the study period of 19 months, 12 cases of cutaneous tuberculosis were recruited for DOTS therapy. All these cases were clinically evaluated with relevant hematological, biochemical and serological tests for human immunodeficiency virus and Venereal Diseases Research Laboratory. In addition, Mantoux test, skin biopsy, X-ray chest and sputum examination were also performed. None of our patients had concomitant pulmonary or other systemic tuberculosis. The diagnosis of cutaneous tuberculosis was established based on clinical picture, histopathology and positive tuberculin test. Before providing category III DOTS, photographs were taken to observe clinical improvement. All these cases were reviewed every week for the first 2 months and, thereafter, every month for 4 months along with clinical photographs to observe clinical improvement on the basis of reduction of size, erythema, induration, scaling and verrucosity. These cases were followed-up to 6 months after completion of DOTS to observe any relapse.
The age group of our patients varied from 6 to 50 years. The different types of cutaneous tuberculosis were: lupus vulgaris (6), tuberculosis verrucosa cutis (2), tuberculids (2) and erythema induratum of Bazins (2). All 12 cases completed 6 months of DOTS therapy and were followed-up to 6 months. In four cases, clinical improvement was very rapid and was observed within 4 weeks, in three cases within 6 weeks and the remaining five cases within 8 weeks. All the cases responded to DOTS during intensive phase. The lesions healed with residual skin changes [[Figure - 1]a,b] and [[Figure - 2]a,b]. The compliance and tolerability with DOTS was good and no side-effects were observed during the DOTS therapy. No relapse was observed during the follow-up period.
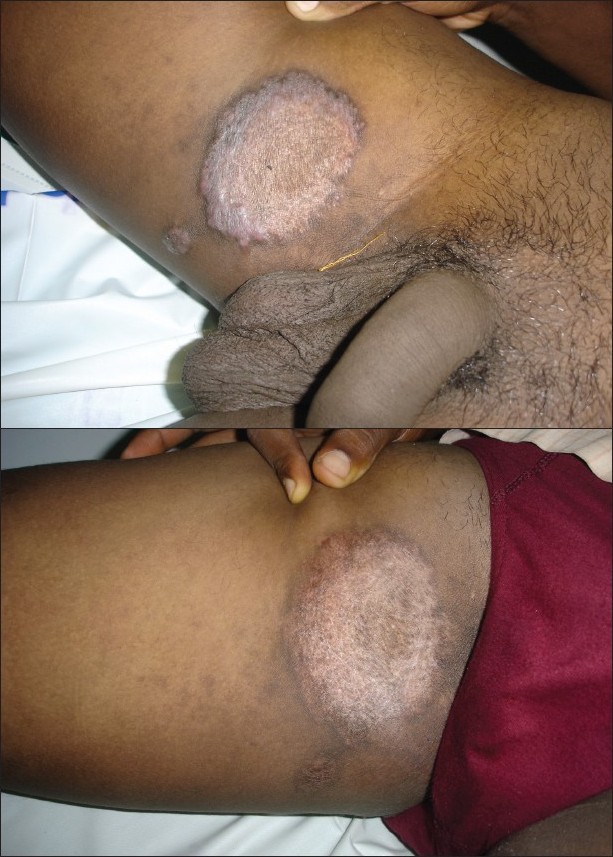 |
| Figure 1: (a) Lupus vulgaris Figure 1b: Lupus vulgaris after 4 weeks of directly observed treatment short course treatment |
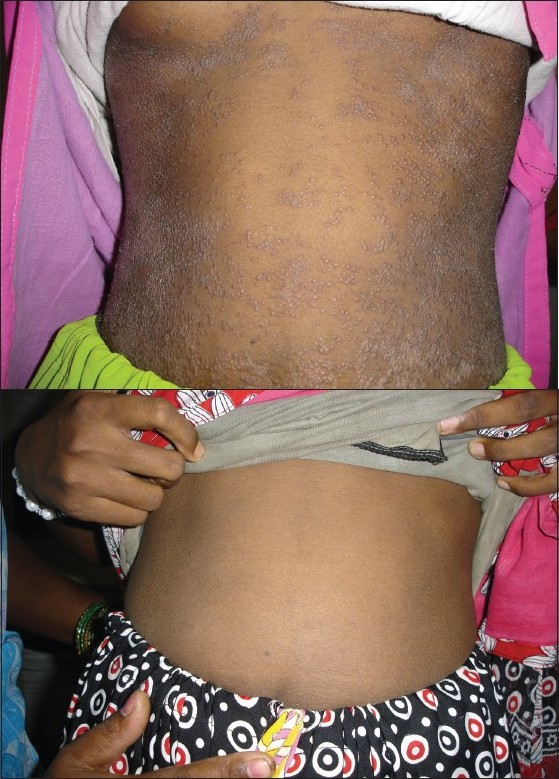 |
| Figure 2: (a)Lichen scrofulosorum Figure 2b: Lichen scrofulosorum after 3 weeks of directly observed treatment short course treatment |
Although there is a variability in the response rates of DOTS in our study, by and large, the response rates are comparable with the standard daily Short Course Chemotherapy. [7] As the majority of cutaneous tuberculosis cases are paucibacillary and are due to hyperimmune response to bacilli, instead of the four drugs-daily regimen, DOTS regimen with three drugs (intermittent therapy) is ideal so that the patient is exposed to less number of drugs. Therefore, the chances of developing drug toxicities and resistance are minimum. In the implementation of the DOTS regimen, DOTS providers are generally available within the vicinity of the patients, thereby minimizing the travails of travel. Moreover, the patient gets the standard drugs under supervision at free of cost. Therefore, the compliance is fairly good. This sample study aims at encouraging the clinicians to take up this national programme so that the benefits of DOTS reaches the deserving. However, large-scale follow-up studies are warranted to establish the efficacy and other aspects of DOTS in cutaneous tuberculosis.
| 1. |
World Health Organisation. Treatment of tuberculosis: Guidelines for national Programmes. WHO; 1993. p. 1-43.
[Google Scholar]
|
| 2. |
RNTCP at a glance. Available from: http://www.tbcindia.org/RNTCP.asp . [Last accessed on 2010 October 5].
[Google Scholar]
|
| 3. |
Arora VK, Rajnish Gupta. Trends of extra-pulmonary tuberculosis under Revised National Tuberculosis Control Programme: A study from South Delhi. Indian J Tuberc 2006;53:77-83.
[Google Scholar]
|
| 4. |
Ramesh V. Sporotrichoid cutaneous tuberculosis. Clin Exp Dermatol 2007;32:680-2.
[Google Scholar]
|
| 5. |
Hassan I, Ahmad M, Masood Q. Lupus vulgaris: An atypical presentation. Indian J Dermatol Venereol Leprol 2010;76:180-1.
[Google Scholar]
|
| 6. |
Behera B, Devi B, Patra N. Mutilating lupus vulgaris of face: An uncommon presentation. Indian J Dermatol Venereol Leprol 2010;76:199-200.
[Google Scholar]
|
| 7. |
Ramam M, Tejasvi T, Manchanda Y, Sharma S, Mittal R. What is the appropriate duration of a therapeutic trial in cutaneous tuberculosis? Further observations. Indian J Dermatol Venereol Leprol 2007;73:243-6.
[Google Scholar]
|
Fulltext Views
1,827
PDF downloads
988





