Translate this page into:
Pericardial effusion with vesiculobullous lesions in a young female
2 Departments of Pathology, INHS Asvini, Mumbai, Maharashtra, India
3 Departments of Medicine, INHS Asvini, Mumbai, Maharashtra, India
Correspondence Address:
R Dhir
Department of Dermatology, INHS Asvini, RC Church, Colaba, Mumbai 400 005, Maharashtra
India
| How to cite this article: Dhir R, Desylva P, Gehi N, Malik A, Singh Y D, Jagannayakulu H, Tampi P S, Ramasethu R. Pericardial effusion with vesiculobullous lesions in a young female. Indian J Dermatol Venereol Leprol 2006;72:175-177 |
 |
 |
 |
 |
 |
 |
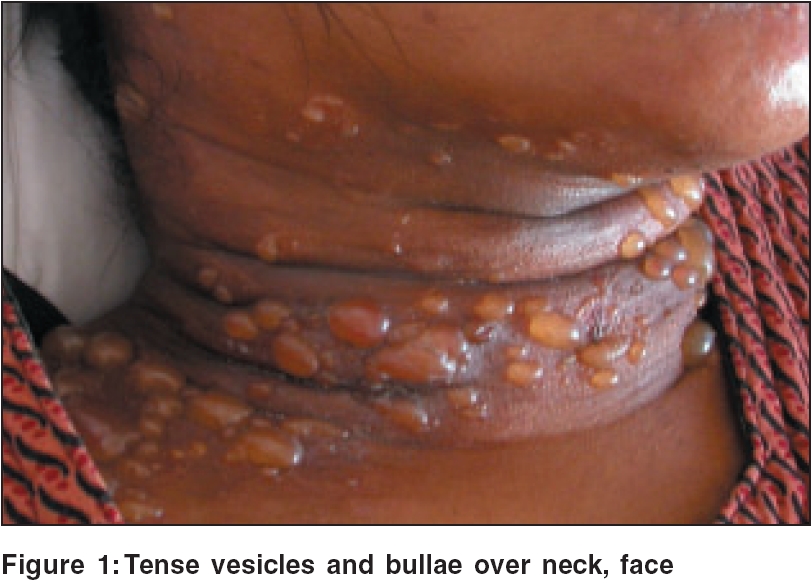
A 27-year-old married lady, resident of Tamil Nadu (South India), was admitted to the ICU with complaints of low grade fever, weakness, dyspnea on accustomed exertion, swelling of face, hands and feet of 15 days duration. Examination revealed tachycardia, blood pressure of 160/100 mmHg, pallor (Hb, 8.2 g%), elevated jugular venous pulse (JVP) and swelling of hands and feet. Imaging of the chest showed pericardial and pleural effusion. Pericardial and pleural taps confirmed exudative pleural and pericardial effusion and she was put on amlodipine, prednisolone 40 mg/day and antitubercular treatment (ATT).
She developed blisters on the skin and mucosal ulcers four days later and was referred to a dermatologist. Skin examination revealed diffuse periorbital edema, multiple tense vesiculobullous lesions on the face, eyelids, neck, trunk and external genitalia [Figure - 1]. Both Nikolskiy and bulla spread signs were negative. Besides, there were multiple painful ulcers on the lips and buccal mucosa. A skin biopsy was done at this stage [Figure - 2][Figure - 3]. ATT was stopped in view of the possibility of drug-induced erythema multiforme. As the 24h urinary protein was markedly elevated (3.5 g/24 h), a kidney biopsy was done. Liver and renal function tests, VDRL, ELISA for HIV, USG abdomen and Mantoux test were within normal limits. IgG and IgM for Mycobacterium tuberculosis were negative. Blood samples were sent for ANA and dsDNA.
What is your diagnosis?
Diagnosis: Bullous systemic lupus erythematosus (bullous SLE) Histopathology of the skin showed necrotic keratinocytes and subepidermal vesicle, which were teeming with neutrophils and a few eosinophils. DIF study revealed IgG deposits at the DEJ. Salt-split studies and Indirect Immunofluorescence (IIF) for type VII collagen were not done because of lack of availability. Kidney biopsy was suggestive of focal proliferative glomerulonephritis (stage III a). Both ANA and dsDNA were strongly positive. The patient was diagnosed as a case of bullous SLE based on the following ACR criteria; oral ulceration, pleural and pericardial effusion (serositis), proteinuria> 3.5 g/day, positive ANA and positive dsDNA.
The dosage of systemic steroids was increased to 60 mg/day. Although the patient had been on systemic corticosteroids for 3 weeks, she still continued to develop multiple fresh blisters. At this stage, she was started on oral dapsone (DDS) 100 mg/day after confirming the glucose-6-phosphate dehydrogenase enzyme level. The blisters started healing in 72 h and the appearance of fresh blisters decreased appreciably. The blood pressure decreased to normal within 2 weeks and antihypertensives were discontinued. Repeat X-ray chest and 2D echo done after 2 weeks did not reveal any pleural or pericardial effusion. The dose of DDS was increased to 150 mg/day as the patient continued to develop few blisters. The patient was discharged on prednisolone 45 mg/day and DDS 150 mg/day. At this stage, the blisters had healed completely, leaving only postinflammatory pigmentary changes. The patient is being followed up every month for the last six months. She continues to be asymptomatic and is on a maintenance dose of prednisolone 15 mg/day and DDS 100 mg/day.
Discussion
SLE is a chronic, recurrent, potentially fatal, multi-system, inflammatory disorder that can often be difficult to diagnose. There is no single diagnostic marker for the disease; instead, it is diagnosed through a combination of clinical and laboratory criteria. Early and accurate diagnosis of this disorder is important because proper treatment can reduce disease morbidity and mortality, especially due to nephritis.[1] The preliminary criteria for the classification of SLE were suggested in 1971 by the American Rheumatism Association (ARA) now known as American College Rheumatology (ACR). These criteria were revised in 1982 using 30 potential variables and later updated in 1997.[1]
Bullous systemic lupus erythematosus is an extremely uncommon subset of lupus erythematosus (LE) and has distinct clinical, histological and immunopathological features. Clinically it manifests as tense vesiculobullous lesions arising upon but not limited to sun-exposed skin. Less than 5% patients of SLE develop vesiculobullous lesions in addition to other cutaneous manifestations like malar rash. However, it is extremely rare to find bullous skin lesions as the only cutaneous manifestation of SLE. The morphology varies from herpetiform vesicles to large hemorrhagic bullae. Apart from a chronic, widespread blistering eruption, a mild, transient blistering eruption has also been reported.[2] The histological examination shows neutrophilic dermal infiltrate and subepidermal separation.[3] Contrary to traditional teaching, IIF studies have shown that type VII collagen may not be the only target antigen in bullous SLE,[4] with Chan et al .[5] showing autoantibodies which labeled both for the epidermal roof and the dermal floor. In fact, certain workers[6] have suggested that there may be two immunologically distinct subtypes of bullous SLE characterized by the presence or absence of antibodies that recognize type VII collagen. Patients of bullous SLE are known to show a dramatic response to DDS.[7]
Pleural and pericardial effusions are by themselves fairly common presentations of SLE.[1] Pericardial effusion may be large in volume but rarely causes tamponade or constriction. It parallels the disease course and the fluid may be serous, serosanguinous or hemorrhagic in nature. However, pericardial effusion as the sole manifestation of SLE is uncommon but not unknown. This combined with the exudative nature of the fluid led to the diagnosis of tuberculosis in our patient. The appearance of tense bullous lesions four days after starting starting ATT further added to the diagnostic dilemma.
The following differential diagnoses were entertained: drug induced EM (which may present only with bullous lesions), bullous SLE, linear IgA dermatoses and epidermolysis bullosa acquista (EBA). Clinically, drug-induced EM was excluded as the patient continued to get vesiculobullous lesions despite stopping ATT. This was further confirmed by histopathology and DIF. The diagnosis of linear IgA dermatosis was excluded as it shows vesiculobullous lesions with annular configuration and deposition of IgA at the DEJ on DIF. The diagnosis of EBA was excluded as it generally shows vesiculobullous lesions on the acral parts, which heal with scars and milia.
Although tuberculosis is very common in a developing country like India, SLE should always be excluded in any multi-system disorder (especially in a young woman). SLE should also be kept in mind in the differential diagnosis of a bullous eruption.
| 1. |
Wojnarowska F, venning VA, Burge SM. Immunobullous diseases. In: Burns T, Breathnach S, Cox N, Griffiths C, editors. Rook's Textbook of dermatology. 7th ed. Blackwell Science: Oxford; 2004. p. 41-53.
[Google Scholar]
|
| 2. |
Fuji K, Fujmoto W, Ueda M, Makino E, Arata J. Detecttion of anti-type VII collagen antibody in Sj φgren's syndrome/lupus erythematosus overlap syndrome with transient bullous systemic lupus erythematosus. Br J Dermatol 1998;139:302-6.
[Google Scholar]
|
| 3. |
Burrows NP, Bhogal BS, Black MM, Rustin MH, Ishida-Yamamoto A, Kirtschig G, et al. Bullous eruption of systemic lupus erythematous: A clinicopathological study of four cases. Br J Dermatol 1993;128:332-8.
[Google Scholar]
|
| 4. |
Yell JA, Allen J, Wajnarowska F, Kirtschig G, Burge SM. Bullous Systemic lupus erythematosus: Revised criteria for diagnosis. Br J Dermatol 1995;132:921-9.
[Google Scholar]
|
| 5. |
Chan LS, Lapiere JC, Chen M, Traczyk T, Mancini AJ, Paller AS, Bullous systemic lupus erythematosus with autoantibodies recognizing multiple skin basement membrane components: Bullous pemphigoid antigen, laminin-5, laminin 6 and type VII collagen. Arch Dermatol 1999;135:569-73.
[Google Scholar]
|
| 6. |
Gammon WR, Briggaman RA. Bullous SL. A phenotypically distinctive but immunologically heterogeneous bullous disorder. J Am Acad Dermatol 1992;27:79-87.
[Google Scholar]
|
| 7. |
Hall RP, Lawley TJ, Smith HR, Katz SI. Bullous eruption of systemic lupus erythematosus: Dramatic response to dapsone therapy. Ann Intern Med 1982;97:165-70.
[Google Scholar]
|
Fulltext Views
1,433
PDF downloads
1,942





