Translate this page into:
Extensive alopecia areata treated with betamethasone oral mini-pulse therapy: An open uncontrolled study
Correspondence Address:
Binod K Khaitan
Department of Dermatology and Venereology, All India Institute of Medical Sciences, New Delhi - 110029
India
| How to cite this article: Khaitan BK, Mittal R, Verma KK. Extensive alopecia areata treated with betamethasone oral mini-pulse therapy: An open uncontrolled study. Indian J Dermatol Venereol Leprol 2004;70:350-353 |
Abstract
BACKGROUND: Extensive alopecia areata is known to respond to daily oral corticosteroids. To minimize the side effects of daily corticosteroids, oral mini-pulse therapy with betamethasone has been used in vitiligo and other dermatoses. There are a few studies in alopecia areata also. AIM: To evaluate the efficacy of oral mini-pulse therapy in extensive alopecia areata. METHODS: It is an open study on sixteen adolescents and adults with alopecia areata/ totalis/universalis treated with oral mini-pulse therapy for a minimum period of six months. The patients were evaluated clinically and with serial photographs for response and periodical investigations were undertaken to look for the side effects. All the patients were followed up for 5-8 months to look for any relapse. RESULTS: Seven (43.7%) patients showed an excellent response and five (31.2%) patients had good response. Two patients (12.5%) had unsatisfactory response and another two (12.5%) were non-responders. There were insignificant / minimal side effects. CONCLUSION: Oral mini-pulse therapy with betamethasone is a safe and effective therapeutic modality for extensive alopecia areata.

 |
 |
 |
 |
 |
 |
 |
 |
INTRODUCTION
Alopecia areata has a variable course and uncertain natural history. All patients of alopecia areata may not require treatment. Spontaneous regrowth in some cases with limited disease process is well known.[1],[2] Although diverse therapeutic options are available, none of them is satisfactory. Moreover, it is not yet identifiable as to which patient of alopecia areata will progress into extensive disease or alopecia totalis/ universalis and will require intervention. In these patients too, the ideal and effective treatment protocol is yet to be evolved.
Systemic corticosteroids have been used in severe forms of alopecia areata.[3] Though their efficacy is beyond doubt, their role in achieving long-term remission is questionable since the drug is required for a long period (several months to 1-2 years) which leads to various side effects. To avoid or minimize the side effects of daily systemic corticosteroids oral mini-pulse (OMP) therapy with various steroids has been used with success in vitiligo,[4] lichen planus,[5] and alopecia areata.[6],[7],[8] We evaluated this regimen of oral mini-pulse (OMP) therapy with betamethasone for extensive alopecia areata/ totalis / universalis in an open uncontrolled study design.
METHODS
Sixteen patients, 11 males and 5 females, between 14-36 years in age (median age 26 years) with severe alopecia areata attending the outpatient department of All India Institute of Medical Sciences, New Delhi were taken for the study. The duration of the disease at the time of presentation ranged from 1 month to 11 years (mean 3 years 2 months). The diagnosis in each patient was made on the basis of history and clinical presentation. Five patients had previously received various modalities of treatment including corticosteroids (systemic daily dose, topical or intralesional), minoxidil and herbal treatment with variable results, while the remaining patients were untreated. However, none of these patients were on any treatment for at least 3 months at the time of their inclusion in the study. There was no history of alopecia areata in the family. Neither was there personal or family history of atopy in any of the patients. All patients were thoroughly evaluated clinically.
The baseline investigations included complete hemogram, blood sugar, serum electrolytes, renal and liver function tests, chest X-ray, urinalysis and examination of stool especially for occult blood. All the patients underwent an ophthalmologic evaluation before starting the therapy. Blood pressure and body weight were recorded initially and monitored every month.
All the patients were treated with oral mini-pulse (OMP) therapy comprising of betamethasone 5 mg given as a single oral dose after breakfast on two consecutive days every week. The patients were treated with this regime for a minimum period of 6 months and then OMP was tapered step-wise every month and stopped in the next 4 months. The patients were evaluated at monthly intervals to assess the therapeutic response and to monitor the side effects of the therapy. Serial clinical photographs were taken every two months to assess the response semi-quantitatively. The investigations were repeated every three months except the chest X-ray which was repeated at 6 months. Hair growth was classified as excellent with 76-100% growth, good with 51-75% and unsatisfactory with < 50% growth. It was considered a treatment failure when there was no growth of hair even after 6 months of treatment. The patients were followed up for 5-8 months after stopping the therapy to look for any relapse.
RESULTS
Nine patients had alopecia universalis and one had alopecia totalis [Table - 1]. The earliest re-growth of hair on the scalp was seen at 1 month. Cosmetically acceptable hair growth was seen at 2-8 months (mean 5 months). This meant that the re-growth covered cosmetically important areas like scalp, eyebrows and in male patients, moustache and beard, showing terminal hair on> 75% of the affected area. The dose of betamethasone was tapered step-wise by 1 mg per dose every month once cosmetically acceptable regrowth was achieved. The eyebrows and eyelashes began to respond in 1-4 months (mean 2 months). Hair growth over the extremities started in about 3-4 months.
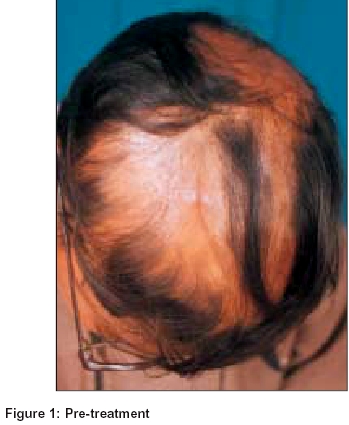
At the end of 6 months, 7 patients (43.7%) showed an excellent response [Figure - 1], [Figure - 2] after which the dose of betamethasone was tapered step-wise and finally stopped. In the subsequent follow-up (5 to 8 months) there was a relapse in 1 patient after 2 months of stoppage of treatment and OMP had to be restarted. Another patient showed scanty re-growth of hair on the eyebrows. Five (31.2%) patients showed a good response after 6 months of therapy [Figure - 3], [Figure - 4]. Betamethasone was continued in these patients in the same doses (5 mg) for another 2 months and then gradually tapered and finally stopped. There has been no relapse in this group of patients. Two (12.5%) patients showed an unsatisfactory response at the end of 6 months. The dosage was tapered in next two months to 3 mg of betamethasone and was continued till one year and then tapered off in the next 3 months. Further improvement was not significant. The remaining two (12.5%) patients were non-responders and both had alopecia universalis. In one of them there was growth of a few vellus hair.
The best response was seen on the scalp. The extremities and eyebrows still showed a few remaining areas of hair loss and required further topical treatment with corticosteroids with variable response. The side effects noted were cushingoid facies and weight gain in 2 patients and acneiform eruption in 2 patients. Three patients complained of mild gastric discomfort which responded to antacids and H2 blockers. At present there are 12 patients who are off treatment since 5 to 8 months and there has been no relapse.
DISCUSSION
The available treatment modalities for alopecia areata can be divided into:[2],[3]
(a) Non-specific irritants - dithranol, phenol
(b) Immune inhibitors - systemic corticosteroids, PUVA
(c) Immune enhancers or universal contact allergens - dinitrochlorobenzene (DNCB), diphencyprone (DPC) and diphenylcyclopropenone (DPCP).
(d) Miscellaneous agents with uncertain mechanism of action and effect -minoxidil.
The efficacy of systemic corticosteroids as a therapeutic modality for severe alopecia areata is observed by various workers but the justification of its use remains debatable. It restores normal hair growth in majority of the cases but most cases have shown relapse at some stage during or after withdrawal of treatment. Pasricha et al[6] treated a single patient with 5 mg of betamethasone twice weekly with a rapid re-growth of hair all over the scalp in 3 months. Sharma[7] administered oral prednisolone as pulses in doses of 300 mg at 4 weeks intervals. Cosmetically acceptable hair growth was seen in 58.3% patients at 4 months of treatment and relapse was seen in 2 patients after stoppage of therapy at 3 and 9 months respectively. In another study, Sharma et al[8] have reported complete hair growth in 26.6% of patients and a good response in 36.6% of patients treated with oral mini-pulse with dexamethasone. In our study, cosmetically acceptable hair growth was seen at a mean of 5 months of therapy. There had been relapse in one patient 4 months after stoppage of treatment who had subsequently been re-started on the same regimen and at present there are 12 out of 16 patients who are off treatment since 5 to 8 months without any relapse.
As per the guidelines of care for alopecia areata[9] after treating a patient with extensive disease for 2-6 months, if no response is seen, another therapy may be tried. Daily oral corticosteroids have high efficacy, however their long-term use may result in serious side effects. To avoid these side effects, pulse therapy was conceived. Dexamethasone-cyclophosphamide pulse therapy in pemphigus patients has been widely accepted.[10] Its chief aim is to provide maximum efficacy with minimal side-effects. With similar intentions, we used OMP with betamethasone in our patients of alopecia areata with equally good results and lesser side effects. OMP may be used for longer periods with minimal side effects as compared to daily doses of corticosteroids. Even if a relapse occurs, the therapy may be repeated and thus remission may be achieved in a larger proportion of cases.
In our study the response was good to excellent in 12 (75%) patients and unsatisfactory or nil in 4 (25%) patients. We therefore conclude that betamethasone oral mini-pulse therapy is a convenient and fairly effective treatment modality for extensive alopecia areata. However, randomized controlled trials with standard therapies on a larger number of patients are required to give more insight into the efficacy and safety of oral mini-pulse therapy for extensive alopecia areata.
| 1. |
Ikeda T. A new classification of alopecia areata. Dermatologica 1965;131:421-45.
[Google Scholar]
|
| 2. |
Montazeri A, Serre G, Kanitakis J. Alopecia areata. Eur J Dermatol 1996;6:471-8.
[Google Scholar]
|
| 3. |
Simpson NB. Alopecia areata. In: Rook A, Dawber R, editors. Diseases of the hair and scalp. 2nd Ed. London: Blackwell Scientific Publications; 1991. p. 324-33.
[Google Scholar]
|
| 4. |
Pasricha JS, Khaitan BK. Oral mini-pulse therapy with betamethasone in vitiligo patients having extensive or fast-spreading disease. Int J Dermatol 1993;32:753-7.
[Google Scholar]
|
| 5. |
Verma KK, Mittal R, Manchanda Y, Khaitan BK. Lichen planus treated with betamethasone oral mini pulse therapy. Indian J Dermatol Venereol Leprol 2000;66:34-5.
[Google Scholar]
|
| 6. |
Pasricha JS, Kumrah L. Alopecia totalis treated with oral mini-pulse (OMP) therapy with betamethasone. Indian J Dermatol Venereol Leprol 1996;62:106-9.
[Google Scholar]
|
| 7. |
Sharma VK. Pulsed administration of corticosteroids in the treatment of alopecia areata. Int J Dermatol 1996;35:133-6.
[Google Scholar]
|
| 8. |
Sharma VK, Gupta S. Twice weekly dexamethasone oral pulse in the treatment of extensive alopecia areata. J Dermatol 1999;26:562-5.
[Google Scholar]
|
| 9. |
Drake LA, Ceilley RI, Cornelison RL, Dobes WL, Dorner W, Goltz RW, et al. Guidelines of care for alopecia areata. J Am Acad Dermatol 1992;26:247-50.
[Google Scholar]
|
| 10. |
Pasricha JS, Khaitan BK, Raman RS, Chandra M. Dexamethasone cyclophosphamide pulse therapy for pemphigus. Int J Dermatol 1995;34:875-82.
[Google Scholar]
|
Fulltext Views
11,710
PDF downloads
1,607





